The purpose of this prospective, single-center study was to measure the value of Krebs von den Lungen-6 (KL-6), a kind of transmembrane mucoprotein, in diagnosing interstitial lung disease (ILD) and in assessing the severity of ILD.
MethodsWe enrolled 184 patients and 30 healthy controls. Ninety-eight patients were diagnosed with ILD, 47 with pneumonia, 19 with non-small cell lung cancer without ILD (NSCLC/non-ILD) and 20 with other lung diseases. Serum KL-6 levels, CT scores of high-resolution computerised tomography (HRCT) and pulmonary function in ILD patients were assessed.
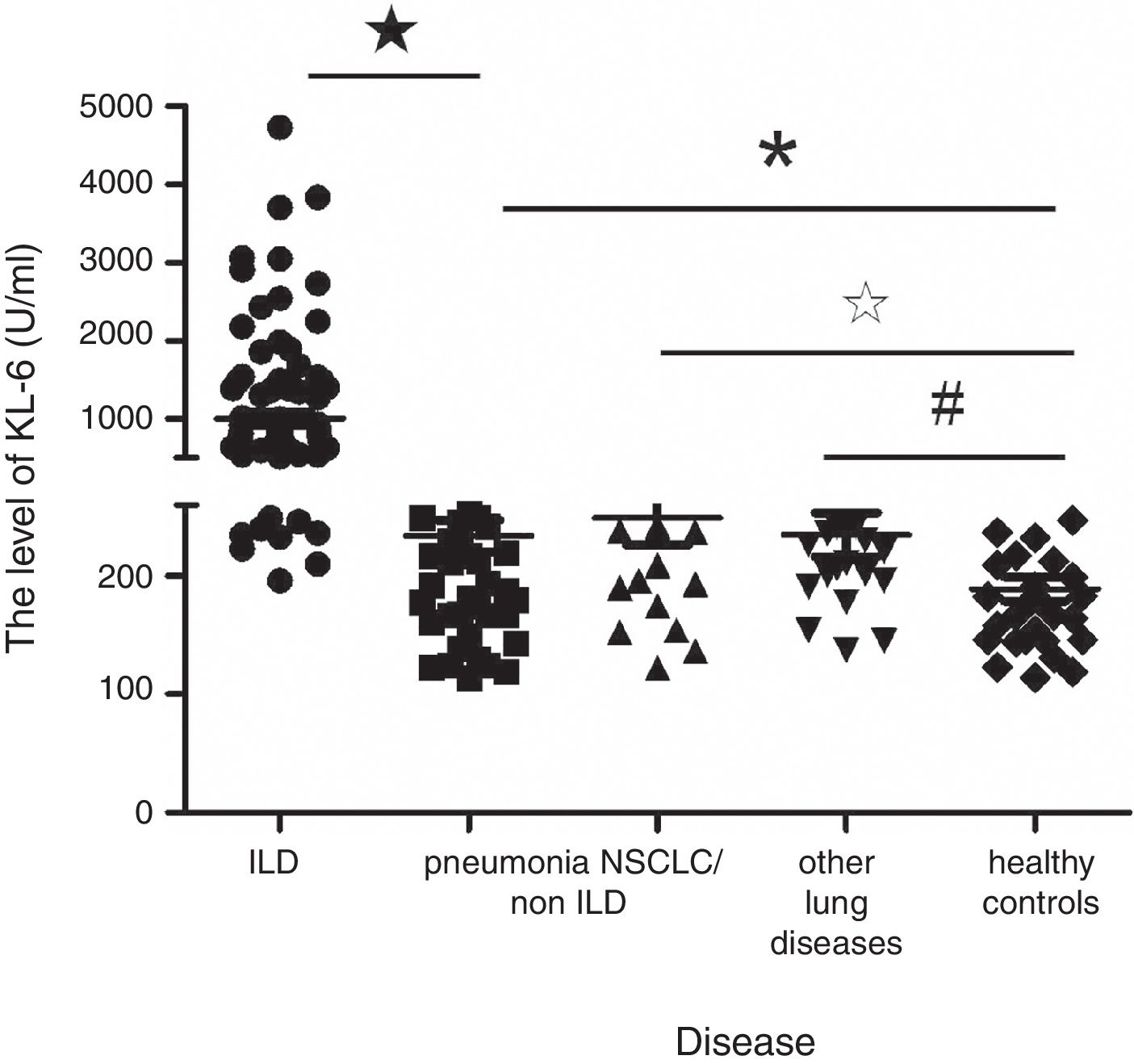
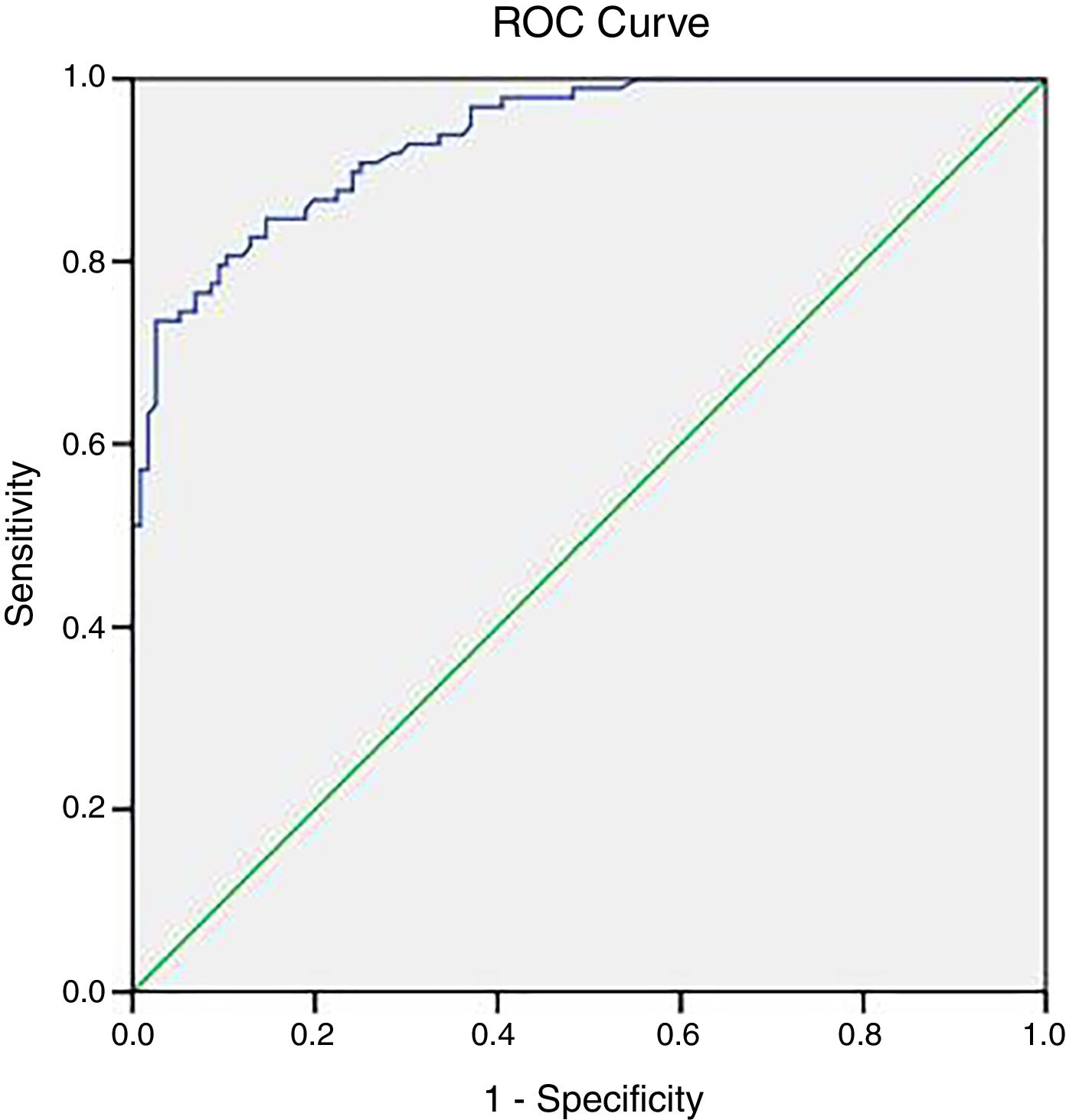
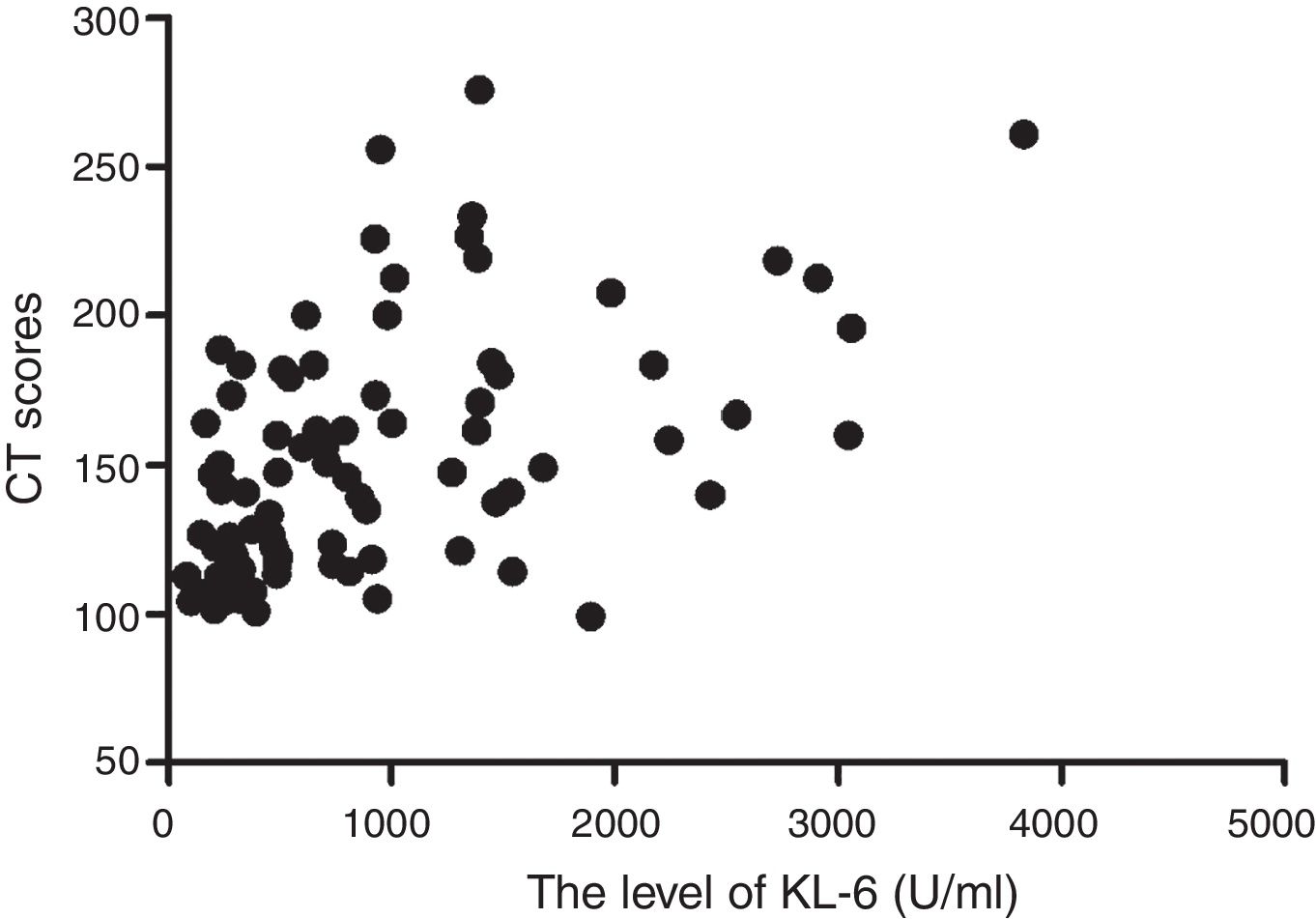
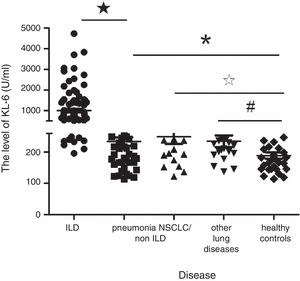
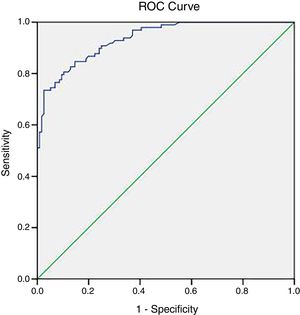
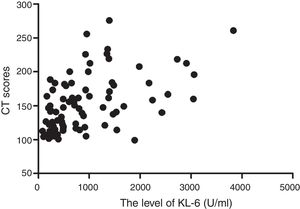
ResultsThe mean value of serum KL-6 in patients with ILD, pneumonia, NSCLC/non-ILD, other lung diseases and healthy controls were 1000.67±882.73U/ml, 234.11±91.02U/ml, 269.95±149.23U/ml, 234.85±83.51U/ml and 189.03±55.50U/ml, respectively. Serum KL-6 levels of patients with ILD were significantly higher than that of other groups (P<0.000). The level of serum KL-6 in patients with pneumonia, NSCLC/non-ILD and other lung diseases was also statistically higher than healthy controls (P<0.05). When the cut-off value was 312U/ml, the sensitivity and specificity of KL-6 for the diagnosis of ILD was 84.7% and 85.3% respectively (AUC: 0.936, 95% CI: 0.906–0.965). The serum KL-6 levels in patients with ILD were significantly positively correlated with the CT scores (r=0.539, P=0.000) and negatively correlated with DLCO (r=−0.513, P=0.000).
ConclusionSerum KL-6 might be useful in the diagnosis of ILD, especially in the hard-to-diagnose cases, with high sensitivity and specificity. Furthermore, KL-6 might be a valuable marker for evaluation of ILD severity.
Interstitial lung disease (ILD) is a group of diffuse lung diseases, mainly involving interstitial tissue, alveoli and bronchioli. This kind of disease includes idiopathic interstitial pneumonia of unknown etiology, autoimmune associated interstitial pneumonia, drug induced interstitial pneumonia, radiation pneumonitis, etc. A diagnosis of ILD can be reasonably based upon typical HRCT appearances and a compatible clinical setting in approximately 70% of cases.1 However, on some occasions, it is difficult to diagnose, causing considerable challenges for the physician to arrange proper treatment for the patients. The clinical course of ILD is highly variable and unpredictable. It can be stable for a long period of time or progress rapidly. Therefore, non-invasive blood biomarker with diagnostic and prognostic potential could assist the diagnosis of ILD, especially in settings with limited medical resources, and would help in the identification of vulnerable patients.
Krebs von den Lungen-6 (KL-6) is a kind of transmembrane mucoprotein,2 with high molecular weight, secreted by proliferating or damaged type II alveolar epithelial cells, which is specific in judging the function of type II alveolar epithelial cells. Previous studies have shown serum KL-6 to be a useful biomarker for diagnosis of ILD.3,4 However, there have been no previous studies that demonstrated that serum KL-6 level has a correlation with the extent of the disease assessed by HRCT score.
We hypothesized that KL-6 might help to distinguish ILD from other lung diseases and the level of KL-6 may correlate with the HRCT scores of ILD. The purpose of this study was to measure the value of KL-6 in distinguishing ILD and other lung diseases and to detect its ability in assessing the severity of ILD.
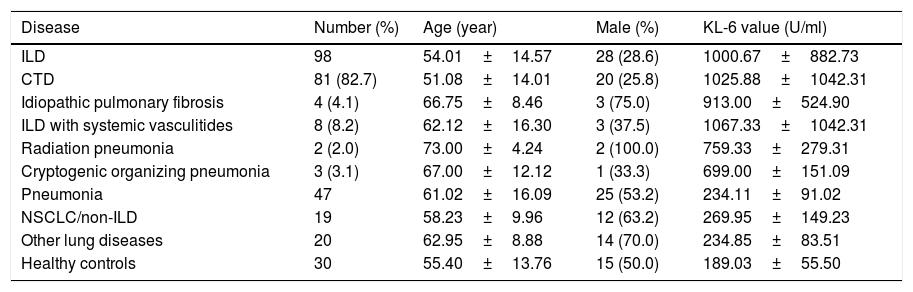
MethodsPatientsA total of 184 patients hospitalized in Renji Hospital, Shanghai, China from July 2016 to December 2016 were enrolled in this prospective, single-center study (Table 1). Ninety-eight patients were diagnosed with ILD, 47 pneumonia, 19 non-small cell lung cancer without ILD (NSCLC/non-ILD) and 20 cases of other lung diseases (14 COPD, 6 asthma). To estimate the group size, we assumed that the sensitivity and specificity of KL-6 in diagnosing ILD was about 85%. With α=0.05, two-tailed and a power of 80%, we needed 77 ILD and non-ILD patients. Considering a compliance rate of 80%, we asked 95 patients to participate in this study. The diagnosis of all patients was based on the guidelines of diseases in China.5 ILD with infection were excluded. In addition, 30 age-matched healthy controls, recruited from participants undergoing a health checkup, with normal pulmonary function test and chest X-ray, and with no history of lung disease were enrolled. The study was undertaken in accordance with the Declaration of Helsinki, and the study protocol was approved by the institutional review board of Renji Hospital (No. 81401411). All patients provided informed written consent for participation in the research and for presentation of the data in this paper.
Basic characteristics of the study population.
| Disease | Number (%) | Age (year) | Male (%) | KL-6 value (U/ml) |
|---|---|---|---|---|
| ILD | 98 | 54.01±14.57 | 28 (28.6) | 1000.67±882.73 |
| CTD | 81 (82.7) | 51.08±14.01 | 20 (25.8) | 1025.88±1042.31 |
| Idiopathic pulmonary fibrosis | 4 (4.1) | 66.75±8.46 | 3 (75.0) | 913.00±524.90 |
| ILD with systemic vasculitides | 8 (8.2) | 62.12±16.30 | 3 (37.5) | 1067.33±1042.31 |
| Radiation pneumonia | 2 (2.0) | 73.00±4.24 | 2 (100.0) | 759.33±279.31 |
| Cryptogenic organizing pneumonia | 3 (3.1) | 67.00±12.12 | 1 (33.3) | 699.00±151.09 |
| Pneumonia | 47 | 61.02±16.09 | 25 (53.2) | 234.11±91.02 |
| NSCLC/non-ILD | 19 | 58.23±9.96 | 12 (63.2) | 269.95±149.23 |
| Other lung diseases | 20 | 62.95±8.88 | 14 (70.0) | 234.85±83.51 |
| Healthy controls | 30 | 55.40±13.76 | 15 (50.0) | 189.03±55.50 |
ILD: interstitial lung disease; CTD: connective tissue disease; NSCLC/non-ILD: non-small cell lung cancer without ILD.
Serum samples of all subjects were frozen at −80°C until assayed. Serum KL-6 levels were measured by latex enhanced turbidimetry immunoassay using SEKISUI kit (SEKISUI medical Co., Ltd., Japan).
HRCT score systemHRCTs of the ILD patients were performed within 24h of taking blood. HRCT data acquisitions were obtained with 1.0–1.5mm at 10-mm intervals at end inspiration from the lung apex to the base. Images were reviewed in random order by two independent radiologists, with 10 and 16 years of experience, who were blind to the diagnosis and clinical course of patients. Each radiologist independently assessed the presence, extent, and distribution of CT findings: ground-glass attenuation (GGA), consolidation, traction bronchiectasis, or bronchiolectasis and honeycombing, as defined by the Fleischner Society,6 and findings were agreed upon by consensus between the two radiologists. The HRCT findings were graded on a one to six scale based on the classification method7,8 as follows: areas with (1) normal attenuation, (2) GGA without traction bronchiectasis or bronchiolectasis, (3) consolidation without traction bronchiectasis or bronchiolectasis, (4) GGA with traction bronchiectasis or bronchiolectasis, (5) consolidation with traction bronchiectasis or bronchiolectasis, and (6) honeycombing.
The lungs were divided into six zones (upper, middle, and lower on both sides); each zone was evaluated separately. The upper lung zone was defined as the area of the lung above the level of the tracheal carina. The lower lung zone was defined as the area of the lung below the level of the inferior pulmonary vein. Lung area between the upper and lower zones was defined as the middle lung zone. Abnormal findings and the extent of lung involvement was evaluated visually and independently for each of the six zones. The score was based on the percentage of the lung parenchyma that showed evidence of the abnormality and was estimated to the nearest 5% of parenchymal involvement.9
Pulmonary function testsAll patients performed tests of spirometry, diffusing capacity and lung volumes within 72h of sampling blood. Maximum forced vital capacity (FVC), forced expiratory volume in 1s (FEV1), vital capacity (VC), diffusing capacity (DLCO) were performed as recommended by the American Thoracic Society.8–13
Statistical analysisDescriptive statistics were used to analyze the data. The measurement data was described as mean±standard deviation. Differences between groups were analyzed by one-way ANOVA. The correlation between different parameters in ILD patients was analyzed by Pearson Bivariate Correlation analysis. Receiver operating characteristic (ROC) curves were used to calculate the sensitivity and specificity. A P<0.05 was considered significant. All data were analyzed by SPSS for Windows, version 16.0.
ResultsClinical data of all casesThe basic characteristics of five groups are shown in Table 1. The frequencies of specific CTDs in ILD group are: 29 dermatomyositis, 15 systemic lupus erythematosus, 3 rheumatoid arthritis, 16 undifferentiated connective tissue disease, 7 polymyositis, 3 Jo-1 syndrome, 4 Sjogren's syndome, 3 scleroderma, and 1 systemic sclerosis. In NSCLC/non-ILD patients, 9 were adenocarcinoma, 10 were squamous cell carcinoma.
Serum KL-6 levelsThe value of serum KL-6 in patients with ILD, pneumonia, NSCLC/non-ILD, other lung diseases and healthy controls were 1000.67±882.73U/ml, 234.11±91.02U/ml, 269.95±149.23U/ml, 234.85±83.51U/ml and 189.03±55.50U/ml, respectively (Fig. 1). Serum KL-6 levels of patients with ILD were much higher than other groups (P<0.000). The levels of serum KL-6 in patients with pneumonia, NSCLC/non-ILD and other lung diseases were also significantly higher than those of healthy controls (P<0.05). However, the serum KL-6 levels between pneumonia, NSCLC/non-ILD and other lung diseases showed no statistical difference (P>0.05). Subgroup analysis in ILD patients showed no difference in the KL-6 serum levels in specific etiologies.
Pulmonary function testing and CT score in ILD patientsIn ILD patients, VC percent predicted, FVC percent predicted, FEV1 percent predicted, DLCO percent predicted and CT score was 73.90±18.77%P, 74.43±19.70%P, 73.94±18.97%P, 47.24±15.39%P, 148.87±41.07%P, respectively.
ROC analysis of serum KL-6 for diagnosing ILDROC curve was analyzed to further investigate the sensitivity and specificity of serum KL-6 levels for the diagnosis of ILD (Fig. 2). According to the area under the ROC curve, when the cut-off value was 312U/ml, the sensitivity and specificity for the diagnosis of ILD was 84.7% and 85.3%, respectively (ACU: 0.936, 95%CI: 0.906–0.965).
To further investigate whether the level of serum KL-6 in ILD patients was related to the severity of pulmonary lesion and function, we analyzed the relationship between serum KL-6 level, HRCT score and pulmonary function in patients with ILD. The results showed that the serum KL-6 levels in patients with ILD were significantly positively correlated with the CT scores (r=0.539, P<0.000) (Fig. 3), and negatively related to DLCO (r=−0.513, P=0.000). HRCT scores and DLCO were negatively correlated (r=−0.534, P=0.000).
DiscussionThe present study demonstrated that KL-6 had a good sensitivity and specificity in diagnosing ILD and the serum level of KL-6 correlated well with HRCT scores and pulmonary function in ILD patients.
ILDs are a group of diseases characterized by interstitial inflammation and fibrosis, and usually have no identifiable cause, so are defined as idiopathic. Those with identifiable causes are usually associated with environmental factors (pneumoconioses), pharmacologic induction (bleomycin or amiodarone), radiation or connective tissue diseases (CTDs). CTD-related ILDs are usually a consequence of the disease process and incidence varies according to the individual CTD. Early and accurate diagnosis of ILD is critical to prevent development from initial inflammatory activity (alveolitis) to end-stage disease with irreversible honeycombing and fibrosis. Standard tests to screen for ILD are pulmonary function tests, chest radiography and HRCT of the lungs. However, these tests are not always sensitive and are sometimes nonspecific.
KL-6 is a kind of transmembrane mucoprotein,2 with high molecular weight. It is secreted by proliferating or damaged type II alveolar epithelial cells, which is specific for judging the function of type II alveolar epithelial cells. The increase secretion or releasing of KL-6 by type II alveolar epithelial cells will be the main cause of the increased serum KL-6 level. In fibrosis lung disease, with the regeneration of type II alveolar epithelial cells, KL-6 level will be greatly increased. The serum KL-6 level has been demonstrated as a sensitivity marker for ILD, such as idiopathic pulmonary fibrosis, hypersensitive pneumonitis and radiation pneumonitis.14 In the present study, it proved, as expected, that there was a positive association between ILD and serum levels of KL-6. The KL-6 had a good sensitivity and specificity in diagnosing ILD. The results of the current study together with those of previous studies15,16 have demonstrated that patients with ILD have higher serum KL-6 levels than those without ILD or a control group. Furthermore, there was no correlation between specific types of CTD and KL-6 levels, making underlying disease-related false positive rates minimal if it is to be considered as a diagnostic tool. Therefore, KL-6 may be used as a sensitive, specific and non-invasive diagnostic test for ILD.
Recent research has shown that serum and bronchoalveolar lavage fluid level of KL-6 has an important value in the diagnosis, treatment assessment and prognosis prediction. It is more valuable than SP-A and SP-D.17–19 We have demonstrated that KL-6 level has a negative correlation with DLCO, which is consistent with the previous studies. Bonella et al.20 reported that serum KL-6 level was increased in 33 cases of pulmonary alveolar proteinosis; the increase of KL-6 was also positively correlated with pulmonary function markers (PaO2, A-aDO2, DLCO, VC and TLC). Higher serum KL-6 levels were associated with worse pulmonary function in these patients. Staples et al.21 found that CT scan scores correlated significantly with clinical and functional severity of interstitial disease. It has been proved that HRCT scores in IPF patients were independently predictive of mortality.9,22 Quantification of the morphologic extent of disease on HRCT has, however, remained difficult to incorporate into routine practice, mainly because of costs and radiation-exposure limit repeatability over time.
To the best of our knowledge, the present study is the first one which proved that serum KL-6 level has a positive correlation with the HRCT score in patients with ILD, indicating that KL-6 might be a valuable marker for assessment of the extent of ILD. KL-6 analysis could greatly reduce the risk of X-ray exposure and is much easier and more acceptable for patients, when used to monitor the therapeutic effect and disease severity.
Previous studies indicated that KL-6 was immunohistochemically detectable in non-small cell lung cancer tissues and the serum KL-6 level was raised in NSCLC patients, which had a prognostic value for those patients. The primary tumor was the origin of circulating KL-6 in NSCLC patients.23 However, it has been proved that compared to ILD patients, the serum KL-6 levels were significantly lower in NSCLC patients, which was in accordance with our results. The present study demonstrated that KL-6 level was significantly lower in NSCLC/non-ILD patients than in ILD patients, although partially overlapping. Furthermore, KL-6 levels showed no significant difference between NSCLC and other lung disease patients. This may be attributed to the small number of NSCLC patients. However, a previous study has demonstrated that elevated KL-6 antigen levels were frequently observed in the sera of patients with lung adenocarcinoma, but infrequently in the sera of those with squamous cell carcinoma.24 In our study, 47.37% patients in NSCLC/non-ILD group were squamous cell cancer, which may partially explain the reason why the KL-6 levels were same between NSCLC/non-ILD and other lung disease patients.
Several limitations to this study warrant comment. The present study did not provide information on the value of KL-6 in prognostication. Further large-scale outcome studies with hard clinical end points will be required to determine the prognostic value of this parameter. However, it has been demonstrated that KL-6 levels correlated well with the morphologic extent of the disease on HRCT and physiological changes measured by pulmonary function test. Even with the highly variable courses of ILDs and different treatment responses, the extent of HRCT changes and baseline pulmonary function results helped to stage the severity of disease and remained the important prognostic factors in ILDs patients. Secondly, the control group was not sex-matched, thus limiting the statistical power and requiring covariance analysis. However, previous studies have shown that there was no difference in KL-6 level by age or gender.25 Furthermore, the historical outcome distinctions between predominantly fibrotic disease and inflammatory disorders (the latter now expanded to include cryptogenic organizing pneumonia, lymphocytic interstitial pneumonia, cellular non-specific interstitial pneumonia, respiratory bronchiolitis with associated interstitial lung disease and desquamative interstitial pneumonia) were exactly as reported previously.1 However, in the absence of biopsy, we could not conclude whether there was any relationship between KL-6 level and pattern of CTD-ILD. Another limitation of this study is the small number of other ILD besides ILD-CTD. However, Zhu et al. showed that there was no difference of the sera level of KL-6 between ILD-CTD and other ILD patients.19
ConclusionIn summary, KL-6 may be a potential new, non-invasive and practicable marker for diagnosis and evaluation of ILD severity. Nevertheless, further prospective clinical studies are needed to define whether levels of KL-6 might have prognostic value in ILD.
Ethical disclosuresProtection of human and animal subjectsThe authors declare that the procedures followed were in accordance with the regulations of the relevant clinical research ethics committee and with those of the Code of Ethics of the World Medical Association (Declaration of Helsinki).
Confidentiality of dataThe authors declare that they have followed the protocols of their work center on the publication of patient data.
Right to privacy and informed consentThe authors have obtained the written informed consent of the patients or subjects mentioned in the article. The corresponding author is in possession of this document.
FundingThis study was funded by Shanghai Natural Science Foundation (14ZR1425200, 16ZR1420600), Chinese National Natural Science Foundation (81401411) and Research Program of Shanghai College Teachers (101005.26.23).
Author contributionsH Qin and HD Jiang conceived this study and supervised all aspects of its implementation; H Qin, XP Xu and J Zou acquired the data; H Qin, XP Xu, J Zou, XJ Zhao and Y Kang analyzed and interpreted the data; H Qin and Y Kang obtained the funding; H Qin, Y Kang and HD Jiang drafted the article and revised critically for important intellectual content. All the authors contributed to the proof reading of the manuscript.
Conflict of interestThe authors have no conflicts of interest to declare.
The authors thank Yunzhe Zhou, Zhikai Zhan and Jiahe Tian for their hard work in word processing.