Coronavirus disease 2019 (COVID-19) is an emerging infectious disease caused by a novel SARS-CoV-2 pathogen. Its capacity for human-to-human transmission through respiratory droplets, coupled with a high-level of population mobility, has resulted in a rapid dissemination worldwide. Healthcare workers have been particularly exposed to the risk of infection and represent a significant proportion of COVID-19 cases in the worst affected regions of Europe.
Like other open airway procedures or aerosol-generating procedures, bronchoscopy poses a significant risk of spreading contaminated droplets, and medical workers must adapt the procedures to ensure safety of both patients and staff. Several recommendation documents were published at the beginning of the pandemic, but as the situation evolves, our thoughts should not only focus on the present, but should also reflect on how we are going to deal with the presence of the virus in the community until there is a vaccine or specific treatment available. It is in this sense that this document aims to guide interventional pulmonology throughout this period, providing a set of recommendations on how to perform bronchoscopy or pleural procedures safely and efficiently.
Coronavirus Disease 2019 (COVID-19), a new infectious disease that emerged in early December 2019 in Wuhan (China),1 is triggered by a novel pathogen with phylogenetic similarity to what caused the severe acute respiratory syndrome (SARS) outbreak in 2003, and was called SARS-CoV-2.2 Its capacity for human-to-human transmission and international air travel facilitated the rapid dissemination on an unprecedented scale to the rest of the world.3,4
In Italy, the latest figures reported that 9% of COVID-19 cases were health care workers (HCW), while in Spain the rate of medical staff infected reached 26%, the highest in Europe.5 In Portugal, by 12th May 2020, 11.3% of infections occurred in HCW.6 There are at least two explanations for such a high number of infected personnel. First, the lack of proper personal protective equipment (PPE) at the beginning of the epidemic, when assisting both confirmed and suspected patients with COVID-19. Second, the duration of exposure to infected patients undergoing aerosol-generating procedures, such as non-invasive ventilation (NIV) and bronchoscopy, directly resulting in a significant increase in the risk of transmission to HCW.
The Portuguese Society of Pulmonology recently issued a set of recommendations for bronchoscopic procedures,7 shortly after the diagnosis of the first cases in Portugal. The document aimed to guarantee the protection of both patients and medical practicioners, and to ensure that the healthcare workforce would be conserved to fullfill their mission throughout the period. Since then, a significant amount of scientific evidence has been accumulated; so, the present document gives an update of the available literature, providing practical suggestions for pulmonologists undergoing bronchoscopy or pleural procedures in the setting of the current and post-pandemic phases.
Risk of transmissionRespiratory droplets comprise the main route of SARS-CoV-2 transmission, although airborne transmission is also possible through aerosol-generating procedures, such as bronchoscopy.8 One study during the H1N1 pandemic provided experimental evidence that bronchoscopic procedures increases more than 4 times the viral copy number per litre in positive air samples.9 While the heavy droplets rapidly settle, aerosol particles are much smaller (<5−10 µm) and are dispersed in the air over extensive distances, posing a considerable risk of infection in enclosed spaces, specially if poorly ventilated.10
The contribution of asymptomatic carriers has also been subject of debate.11,12 A significant proportion of them have lung abnormalities on chest CT scans13,14 and a high level of viral shedding may be detected in presymptomatic patients,14 so it is likely that transmission occurs in the early stages of infection when patients are either minimally symptomatic or asymptomatic. Unrecognized patients pose a real challenge to infection control and, when not promptly handled with appropriate airborne precautions, are one of the most critical factors for SARS-CoV-2 infection spread in the healthcare setting.
MethodsThe Portuguese Pulmonology Society appointed FG to chair this consensus group. Seven national IP specialists were selected based on their clinical expertise and different settings (university vs. non-university hospitals; state vs. private hospitals; pulmonologists vs. critical care specialists; ...). At the first online consensus meeting, attended by all members, a primary draft with several sections was created. This was shared online and further improved by written comments and suggestions. Then, each IP specialist was assigned a specific section presented in this document and was responsible for reviewing and evaluating the relevant available literature related to the topic. Electronic databases (Pubmed, OVID Medline and Embase, Web of Science, Cochrane Central Register of Controlled Trials) were used to search for the terms “COVID-19” OR “SARS-CoV-2” AND (“bronchoscopy” OR “interventional pulmology” OR “thoracentesis” OR “thoracocentesis” OR “pleural effusion” OR “pneumothorax” OR “rigid bronchoscopy” OR “thoracoscopy” OR “chest drain”). Position papers from major health organizations (US Centers for Disease Control and Prevention, European Centre for Disease Prevention and Control and World Health Organization) and important scientific societies (European Respiratory Society, European Association for Bronchology and Interventional Pulmonology, American Association for Bronchology and Interventional Pulmonology, World Association for Bronchology and Interventional Pulmonology and British Thoracic Society) were also reviewed.
In a second online conference the complete draft was evaluted by all team members and two working groups were created. They were responsible for discussing and revising different sections, and editing the text for consistency. Afterwards, the final manuscript was distributed to the consensus group members and assessed for final approval.
Adaptations of the interventional pulmonology (IP) departmentAlthough there is still some heterogeneity in the definition and scope of of “interventional pulmonology” (IP), it has become the most widely accepted term to describe the use of techniques for the diagnosis and treatment of a growing number of thoracic disorders.15
In the context of this document, the term IP is used to encompass the concepts of bronchoscopy (diagnostic or therapeutic), advanced bronchoscopy (flexible or rigid bronchoscopy and all its associated techniques), pleuroscopy (rigid or semi-flexible) and other simpler pleural techniques (such as thoracentesis, placement of thoracic drainage systems and indwelling pleural catheters). Though we acknowledge this wider definition of IP may be controversial, it covers all technical domains that most Portuguese pulmonologists need to address, and for the purpose of this document, it positions us to issue general recommendations. In the following subsections, specific scenarios of different technical specializations will be addressed in order to overcome this broader definition and to apply it better to individual settings.
The IP department is a high-risk area, given the type of procedures that are performed with airway manipulation and with multiple staff involved. Although this setting is generally designed to deal with occasional airborne infectious diseases, such as tuberculosis, it is not prepared to systematically assess high-risk cases that need additional resources, diminish productivity and effectiveness and generate a huge workload.
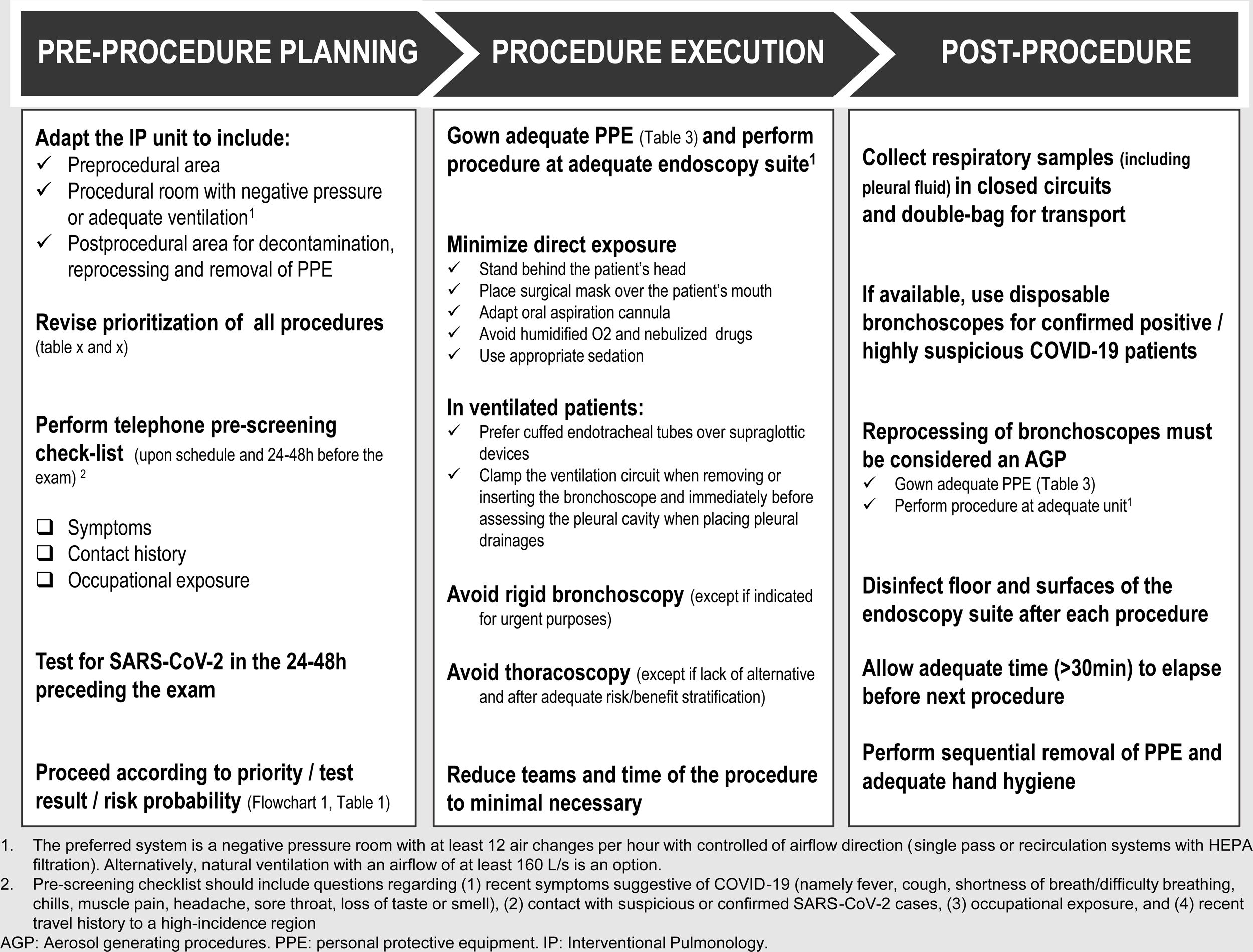
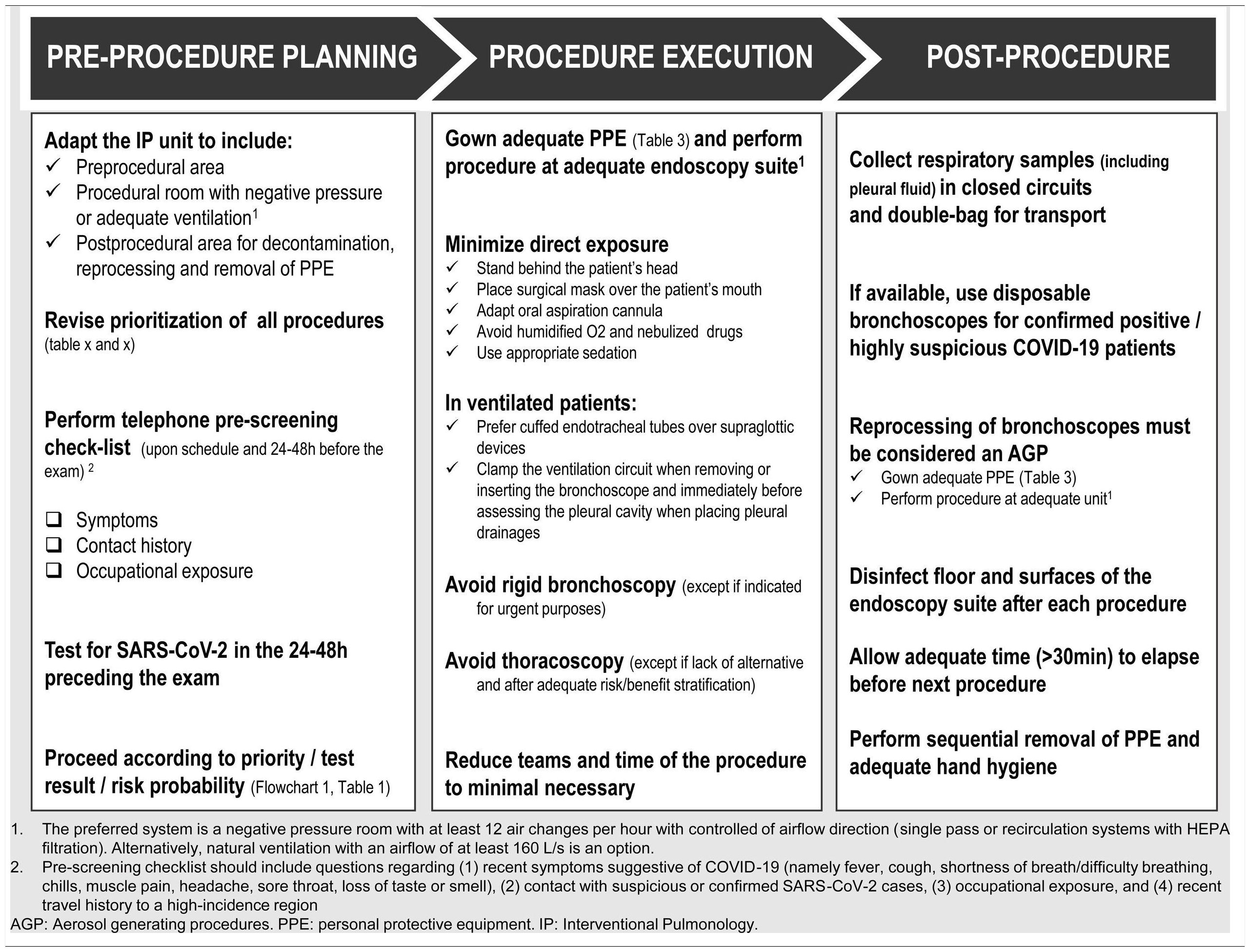
Thus, each IP unit must rethink their administrative and logistic circuits in different areas, as well as the type and timing of performed procedures, to protect both HCW and patients. Moreover, as international health associations advocate, an infection-control program in healthcare settings should be implemented, consisting of a three-level hierarchy, including administrative, environmental and engineering controls, and personal protection equipment (PPE).16 In the following subsections, each of the above listed hierachic levels are briefly presented.
Administrative and organizational issuesAdministrative and logistic measures are crucial to ensuring safety while still maintaining IP activity.17 Some general precautions include:
- -
All referrals and requests to the IP unit must preferably be made by telephone or digital means.
- -
Upon schedule and 24−48 h prior to arrival at the IP Unit, patients should be contacted by telephone and submitted to a pre-screening checklist that includes questions about 1) recent symptoms suggestive of COVID-19 (e.g. fever, cough, chills, muscle pain, shortness of breath/difficulty breathing, headache, sore throat, loss of taste or smell); 2) contact with suspicious/confirmed SARS-CoV-2 cases; and 3) occupational exposure.
- -
Patients who have recent respiratory and infectious symptoms and/or chest imaging suggestive of COVID-19, should have their elective procedures postponed and rescheduled after all symptoms are resolved.
- -
On arrival at the IP Unit, all patients must be asked again for respiratory symptoms and have their temperature checked.
- -
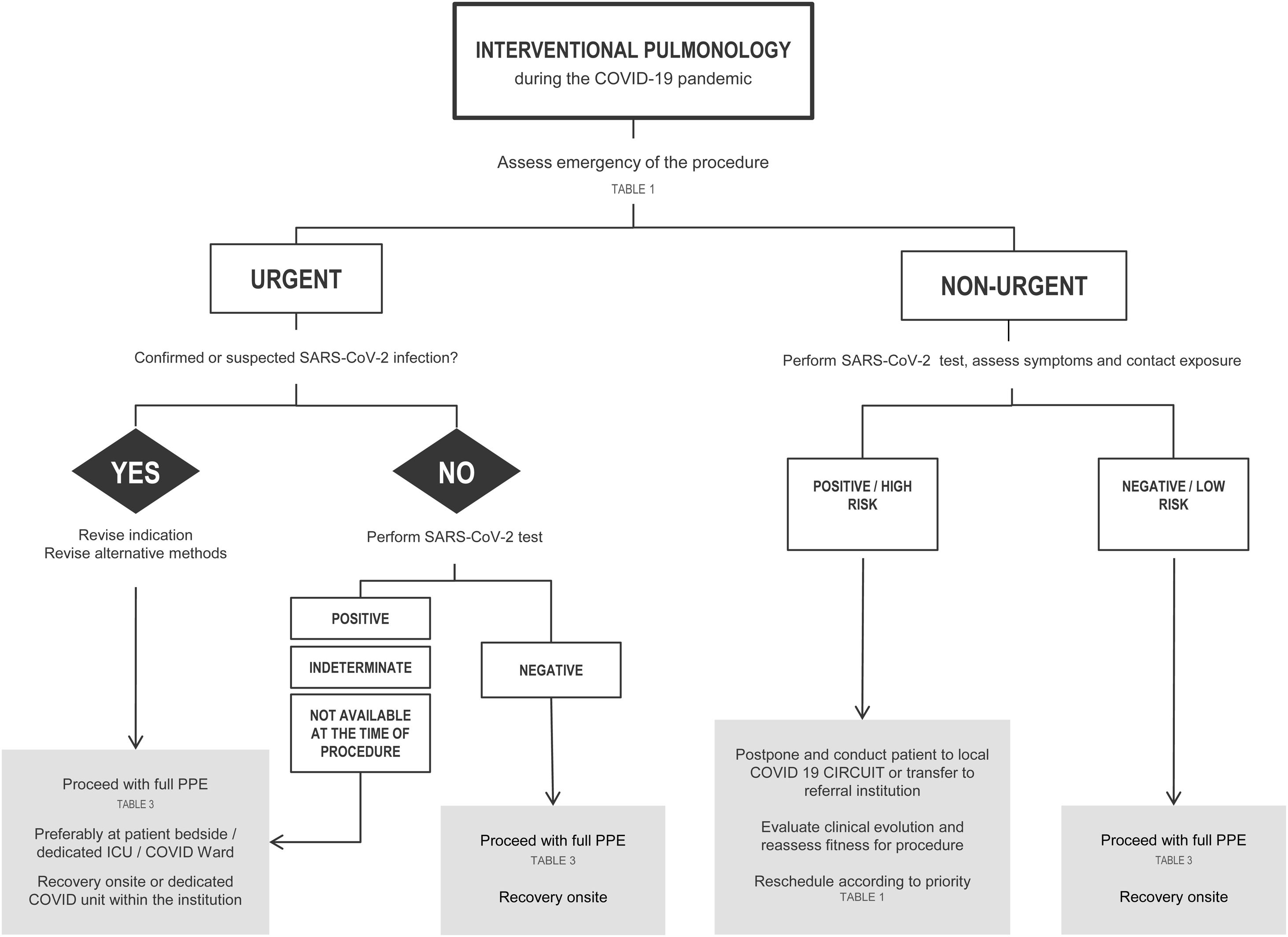
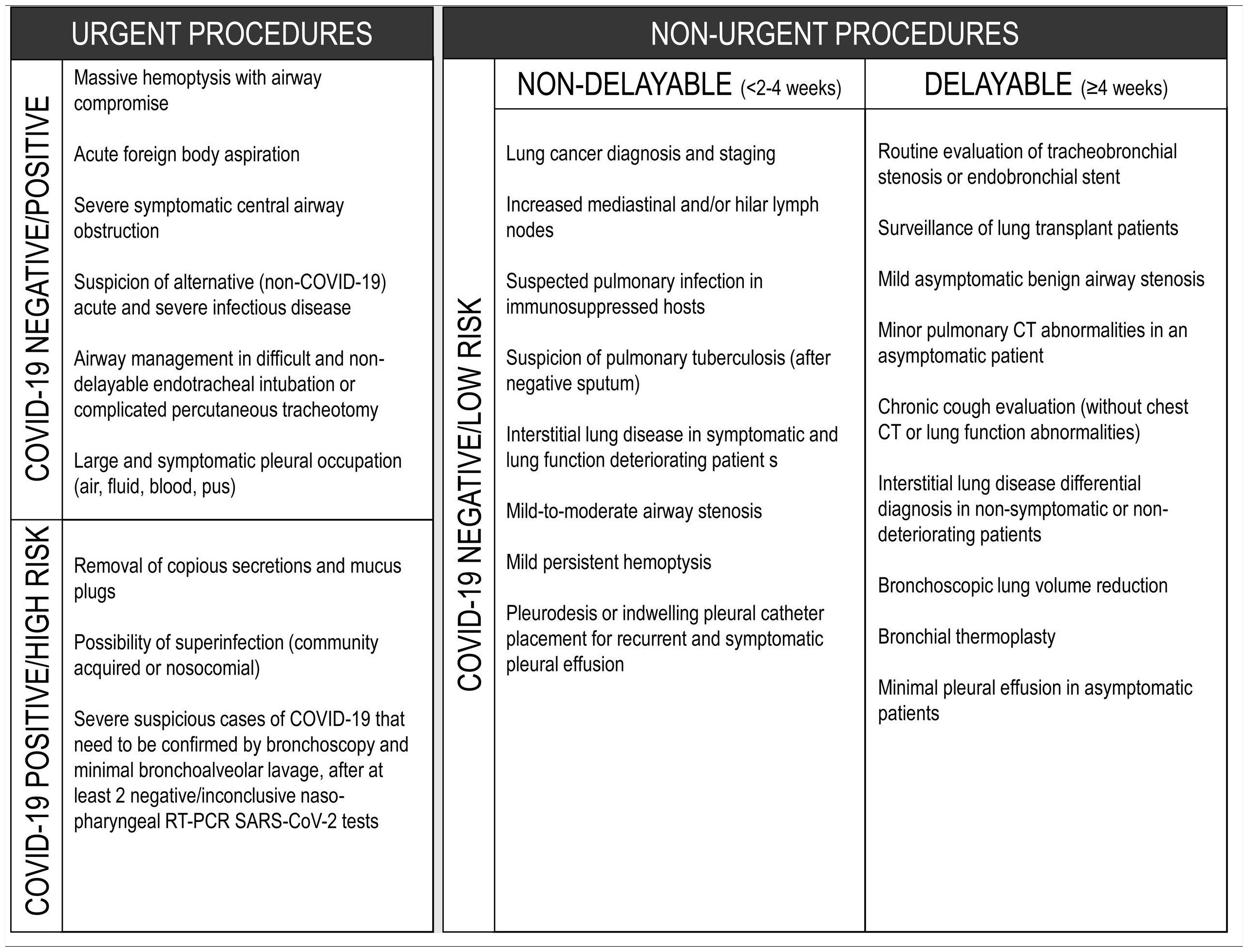
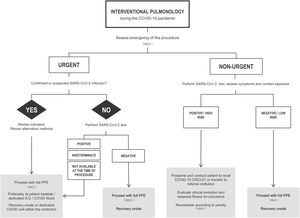
If possible, all patients should have at least one negative RT-PCR for SARS-CoV-2 in the 24−48 h preceding the exam. In patients with a positive RT-PCR SARS-CoV-2, the decision to procede with the intervention will be based on the urgency of the procedure (Chart 1 and Table 1).
- -
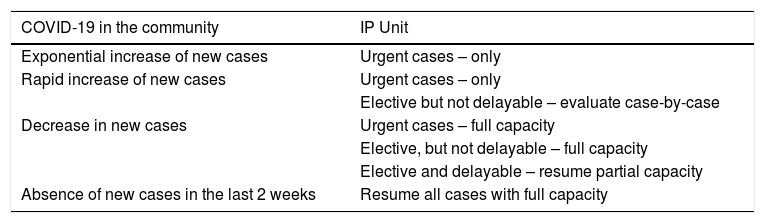
The IP unit should keep a record of deferred patients to reschedule their procedures according to the COVID-19 outbreak situation, as proposed in Table 2.
Table 2.Schedule of IP procedures according to the stage of COVID-19 pandemic.
COVID-19 in the community IP Unit Exponential increase of new cases Urgent cases – only Rapid increase of new cases Urgent cases – only Elective but not delayable – evaluate case-by-case Decrease in new cases Urgent cases – full capacity Elective, but not delayable – full capacity Elective and delayable – resume partial capacity Absence of new cases in the last 2 weeks Resume all cases with full capacity
The design of strategies to minimize risks and a protocol fitting the characteristics of each specific Unit are crucial.18
- -
Reception, administrative, clinical and waiting areas should separate confirmed/high-risk patients from negative/low-risk ones. In addition, inpatients should be segregated from outpatients, either by time or physical location, to prevent cross infection.
- -
Specific circuits and written workflow plans must be prepared, covering the pre-procedural area, procedural room, post-procedural area, decontamination and reprocessing. The implementation of a flowchart with different areas and walking paths using a visual colour zone system can be useful: 1) red zone for contaminated areas; 2) yellow zone for transition areas, and 3) green zone for non-COVID-19 safe areas19 (Fig. 1A). These need to be formulated by internal elements from the IP unit with the cooperation of a multidisciplinary team of hospital members, including administration, engineers, and infection control board.
Figure 1.A. Implementation of specific circuits with colour visual zone system to distinguish contaminated (1, red zone), transition (2, yellow zone) and safe cleaned areas (3, green zone). B. Designated area for donning and doffing of PPE, where posters and other visual aids were placed strategically to act as reminders.
- -
A specific place to store and retrieve all items required for PPE should be defined inside the Unit.
- -
A designated area in the Unit should be selected, close to the procedural suite, for gowning and removal of all PPE, according to hospital protocol and standards, in order to reduce exposure to contaminated particles and droplets. When an anteroom is available, it may be used as an area for donning and doffing of PPE (Fig. 1B).
- -
Stations should be created to facilitate frequent hand hygiene and to distribute waste containers according to local infectious control recommendations. Posters and other visual aids should be placed at strategic locations around the intervention suite to act as reminders.
- -
Emergency procedures in COVID-19 positive patients should preferably be performed within the ICU enviroment, with controlled airway through cuffed endotracheal tube and assisted ventilation.
- -
Elective procedures should be reserved for COVID-19 negative patients (Chart 1 and Table 1). Nevertheless, these procedures should still be performed in a dedicated negative pressure room (see below, ventilation requirement) with strict isolation precautions and sufficient ventilation to avoid aerosol contamination.20 If these requirements are not met in the bronchoscopy suite, then in a different venue, such as an operating theatre, isolation room or the ICU with negative pressure, if available.
- -
If negative pressure rooms are unavailable througout the instituition, a specific and dedicated room with adequate natural ventilation (see requirement below) may be an alternative, provided that appropriate intervals between procedures are reserved and that the suspected COVID-19 cases be programmed after all planned non-COVID daily activity, so that the unit can be carefully cleaned (following the disinfection policy) and ventilated.
- -
Keep the endoscopy room for procedures only (all other activities, such as planning, reporting and laboratory requisition should take place elsewhere).
- -
Suspected and confirmed cases of COVID-19 must be placed in an airborne infection isolation room with negative pressure before and after the procedure. Low-risk and negative patients can remain in the pre-procedural and recovery area, if there is adequate room ventilation, protective equipment (e.g. surgical mask) and physical distance (>2 m) from other negative patients.
- -
Patient source control strategies, such as wearing a mask should be encouraged.
- -
Whenever feasible, it is recommended procedures are pereformed in a room that meets the ventilation requirements for Airborne Infection Isolation (AII), ensuring the dilution and removal of contaminated air. The preferred system is a negative pressure room with at least 12 air changes per hour (ACH) with airflow direction control (single-pass or recirculation systems with HEPA filtration). Alternatively, natural ventilation with an airflow of at least 160 L/s is an option.19,21
- -
Enough time should be allowed to ensure that contaminated air is removed from the room before performing another procedure in the same room (depending on ACH and disinfection methods, but at least 30 min). Local adaptations must be considered according to the characteristics of the IP unit.
Endoscopes are considered semi-critical medical instruments according to the Spaulding classification.22 Recommendations from the Centers for Disease Control and Prevention (CDC) on reprocessing procedures should be followed. These include pre-cleaning, leak-testing, manual cleaning and visual inspection followed by disinfection/sterilization.
- -
A high-level manual disinfection or using an automated endoscope reprocessor is recommended.
- -
Proper storage and documentation are also an integral part of the reprocessing workflow.
- -
A pathway of contaminated equipment must be defined, as well as adequate packaging to minimize exposure (for example, a hermetic box).
- -
If available, disposable bronchoscopes are recommended for confirmed COVID-19 patients with clear advantages in portability, post-procedural handling and cross contamination risk.23
- -
Floors and surfaces of the endoscopy suite must be disinfected after each procedure.
- -
Intermediate level disinfectants with proven activity against enveloped viruses include 0.1% sodium hipochlorite, 62–71% ethanol, 0.5% hydrogen peroxide and quaternary ammonium compounds.19,24,25
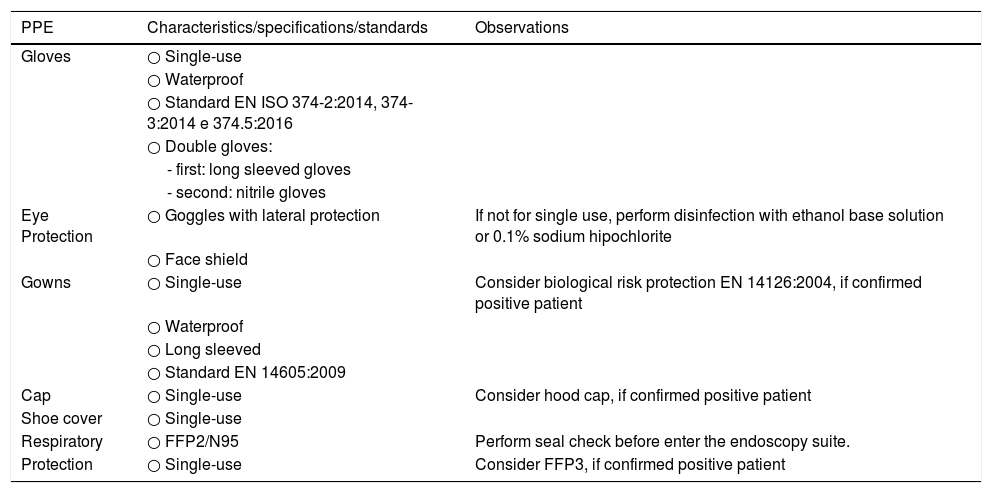
IP procedures are considered to be consistently subjected to the highest risk of exposure. In this setting, full precautions must be taken to cover all different possible types of transmission (contact, droplet and airborne).26 Personnel involved in the reprocessing procedure must also wear protective equipment consisting of eye protection, respiratory mask FP2, long sleeved gown and double gloves.27,28 The recommendations for the use of PPE are shown in Table 3.
Specifications for personal protective equipment during IP procedures.
| PPE | Characteristics/specifications/standards | Observations |
|---|---|---|
| Gloves | ○ Single-use | |
| ○ Waterproof | ||
| ○ Standard EN ISO 374-2:2014, 374-3:2014 e 374.5:2016 | ||
| ○ Double gloves: | ||
| - first: long sleeved gloves | ||
| - second: nitrile gloves | ||
| Eye Protection | ○ Goggles with lateral protection | If not for single use, perform disinfection with ethanol base solution or 0.1% sodium hipochlorite |
| ○ Face shield | ||
| Gowns | ○ Single-use | Consider biological risk protection EN 14126:2004, if confirmed positive patient |
| ○ Waterproof | ||
| ○ Long sleeved | ||
| ○ Standard EN 14605:2009 | ||
| Cap | ○ Single-use | Consider hood cap, if confirmed positive patient |
| Shoe cover | ○ Single-use | |
| Respiratory | ○ FFP2/N95 | Perform seal check before enter the endoscopy suite. |
| Protection | ○ Single-use | Consider FFP3, if confirmed positive patient |
PPE, personal protective equipment.
Samples from the upper and lower respiratory tract, including pleural effusion, are deemed to be the most potentially infectious. Consequently, they should be handled as Category 3 pathogen and double-bagged (first the specimen must be bagged in the patient’s room and then taken out of the room and placed in a separate pre-labeled specimen bag). All specimens must be manually delivered.28,29
Safety rules for staff and patientsIt is also important to define proper new rules for both HCW and patients circulating in the IP unit, as listed below:
Health professionals- -
The IP Unit should reduce and prioritise the allocation of human resources according to the outbreak evolution and hospital needs. The minimum number of staff required to ensure a correct operation must be clearly defined.
- -
It is essential that all personnel follow, train and maintain competency in effective hand hygiene and every aspect of PPE (theoretical, training and simulation sessions) so that everyone is familiar with their role.
- -
All interactions with patients, including informed consent, should be done with appropriate PPE and frequent hand washing. The staff should not reduce the level of awareness and protection, and the idea that patients with suspected COVID-19 should be handled in the same manner as confirmed cases must be reinforced.
- -
A core team that includes only essential HCW should perform the procedure on SARS-CoV-2 positive patients. The most experienced staff should be responsible for the exam to reduce time and deal effectively with possible complications. Other healthcare personnel, such as residents, medical students and visitors should not be inside the unit and the examination room before, during or after the procedure.
- -
Of note, the scheduled exams must be done during normal working hours (avoiding an emergency basis or setting) and in an appropriate, designated room that fulfils all the standards required for care.
- -
Respiratory and contact isolation should be standard and mandatory for all patients. Outpatients and inpatients should always enter the IP Unit with a suitable face mask and keep it on at all times (until the beginning and after the end of the procedure) to minimize the risk of transmission. No unnecessary personal items should be brought into the IP unit.
- -
Family members and caregivers should not stay in the IP waiting rooms. In case of children or patients in need of support, the Unit can allow a single relative to enter the preparation area to provide aid.
- -
The entry into the Unit of suppliers and medical devices sales representatives must be restricted.
Scheduled elective procedures should be reviewed and cancelled if potentially delayable, until local control of the outbreak is achieved. After flattening the infectious curve, many elective IP procedures will have to be performed, as they are essential to provide a definitive diagnosis and effective treatment. At this time, it is advisable to evaluate the delayed requests and to optimize the procedure planning based on clinical needs and operational capability.
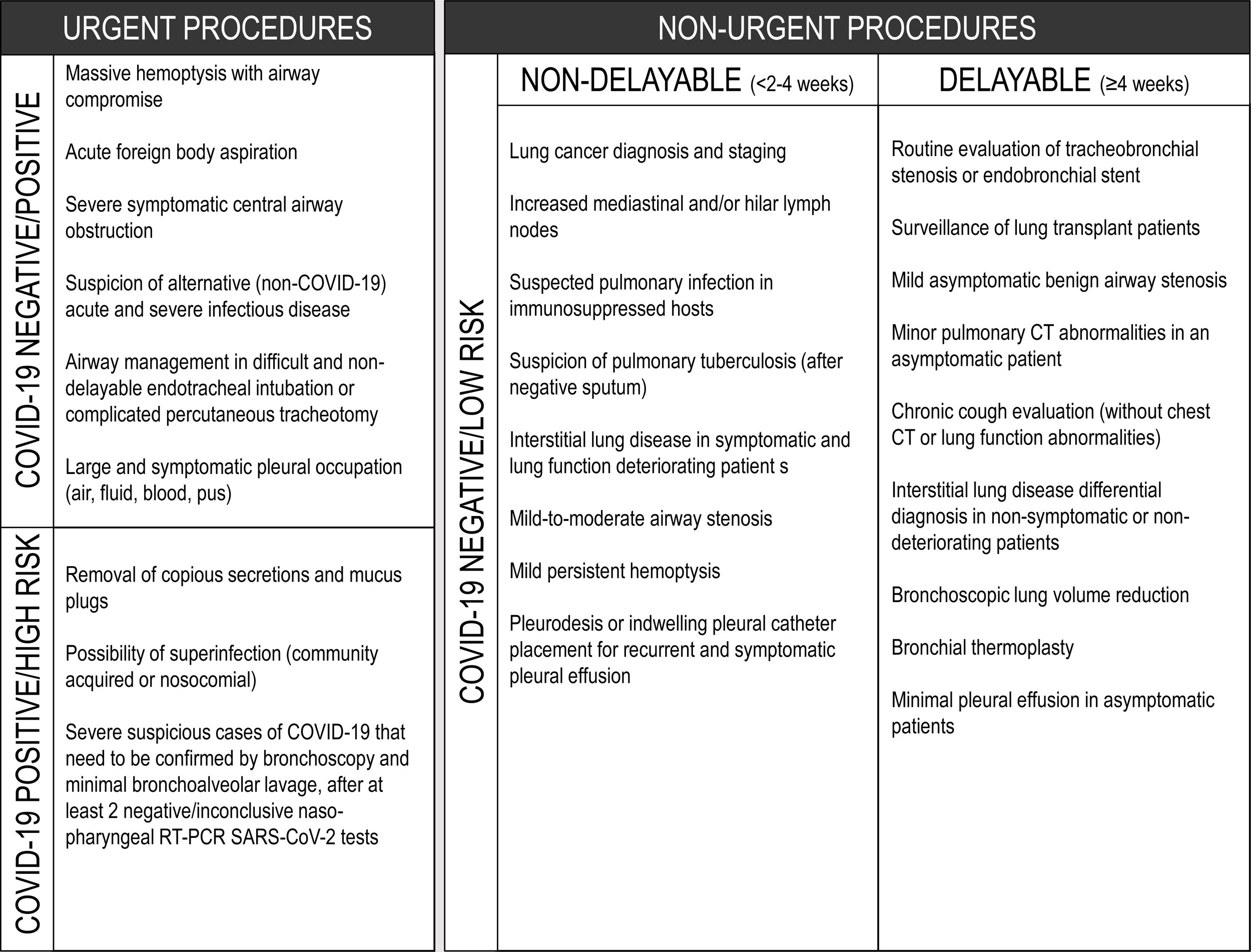
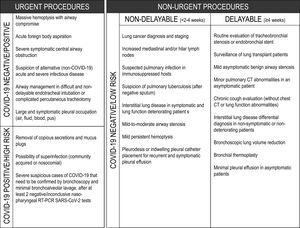
A suggested rational approach for stratification of procedures is provided in Table 1, but we recognize that, in certain cases, the indication may not be straightforward, and the risk-benefit must be weighted on an individual basis by the IP team.31 Although rescheduling certain procedures is obvious in other cases it may not be desirable or ethical. It is important to note that these indications may change according to local epidemiological conditions and the response capabilities of the healthcare system. Several societies have recommended different levels of procedure stratification.26,30,31 Briefly, what is recommended is a stepwise reopening of elective IP procedures according to the national and local COVID-19 outbreak situation, depending on the number of new confirmed cases, hospital admitted cases (ward and ICU), availability of equipment and healthcare staff, time elapse and number of postponed IP cases. Some authors34 have proposed a summary of the elective endoscopic procedure by phases, as shown in Table 2. Anyway, it should be noted that the evolving procedural criteria should always be communicated to other physicians who refer patients for invasive respiratory procedures and to the hospital administration.
Recommendations for bronchoscopyBronchoscopy under spontaneous ventilationThe following reccomendations are expert opinion-based and should be adapted to local regulations and guidelines. In an optimal scenario, it is safer to perform elective bronchoscopy under general anesthesia and orotracheal intubation, clinical conditions permitting. If this is not possible, bronchoscopy can be performed under spontaneous ventilation. Some recommendations are listed below:
- -
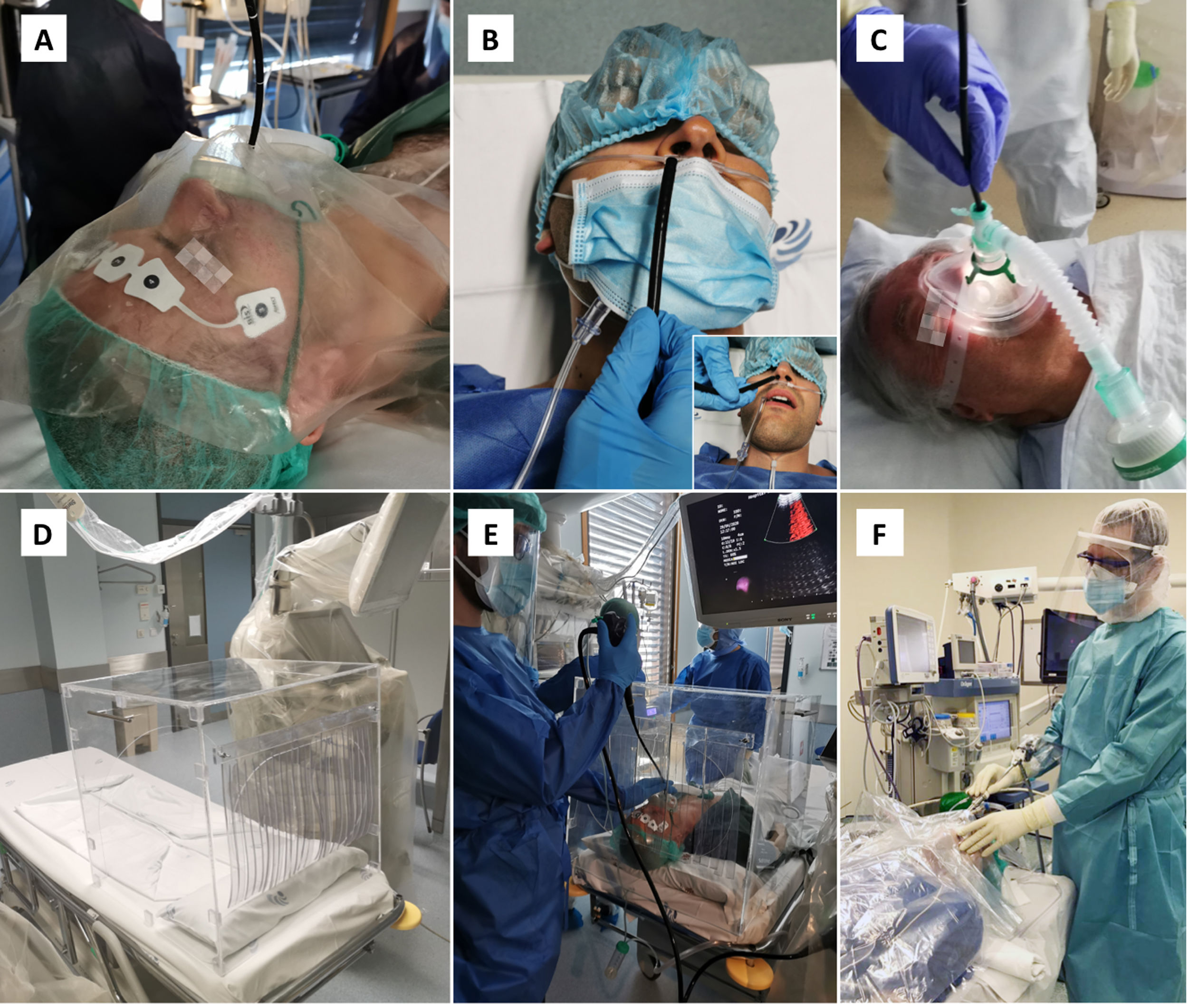
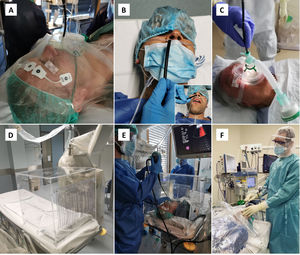
Operator should be standing behind the patient’s head to reduce direct exposure. Oxygen supplementation should be done without the use of humidification, either through a nasal cannula or preferably with an oxygen mask with an entrance to the bronchoscope (Fig. 2A).
Figure 2.Strategies to minimize droplets dispersal during bronchoscopy. A. The bronchoscope may be introduced through an opening made at the oxygen mask, in this case with an additional plastic sheet covering the patient’s head. B. Transnasal approach, with oxygen supplementation through nasal cannula and a surgical mask placed over the patient’s mouth and the oral aspiration canulla. C. Bronchoscopy can be performed under ventilatory support, using a closed circuit ventilation and non-ventilated masks with a dedicated bronchoscope entrance. D. Transparent protective box may contain droplet particles inside. E. Protective box placed over the patient’s head during endobronchial ultrasound. F. Rigid bronchoscopy with rubber caps on the ports of the scope and a plastic covering.
- -
For flexible bronchoscopy, a transnasal approach should be preferred, and a surgical mask should be placed over the patient’s mouth to minimize droplet emission (Fig. 2B).
- -
In hypoxemic patients, bronchoscopy can be performed under NIV, using a closed circuit ventilation (double circuit with viral filters in both arms) and non-ventilated masks with a dedicated bronchoscope entrance (Fig. 2C). High performance NIV ventilators with FiO2 regulation are preferable. From the end of the procedure, NIV should be continued for 1−2 h, titrating the FiO2 to obtain an SpO2 of around 94–95%.
- -
Bronchoscopy under nasal high-flow oxygen therapy is not reccomended and thus should be avoided.
- -
Nebulized medications should be avoided before or after the procedure.
- -
Proper sedation should be used to minimize cough reflex and to increase patient cooperation.
- -
An oral aspiration cannula should be available during the procedure (Fig. 2B).
- -
A transparent protective box may enhance safety by containing dispersal of droplet particles (Fig. 2D). The box is placed over the patient’s head prior to bronchoscopy, with the anesthesia equipment already in place. The bronchoscope is inserted through the covered opening behind the patient (Fig. 2E).
The following recommendations are directed for patients under mechanical ventilation in an ICU setting due to respiratory failure. As reported, 5% of COVID-19 patients can develop respiratory failure and will need ventilatory support32; moreover, associated bacterial, viral and fungal co-infection should not be negleted.33 In critically ill patients under invasive ventilation, ventilator-associated pneumonia occurs in up to 30% and lobar collapse is frequent and multifactorial.34 The same adaptations apply to elective procedures under general anesthesia, performed in the Bronchoscopy Unit or Operating Theatre.
- -
A cuffed endotracheal tube is preferred over supraglotic devices, such as a laryngeal mask; cuff pressure should be maintained between 25–30 cmH2O.35
- -
General anesthesia with muscle relaxant is recommended to reduce the aerosol production.
- -
FiO2 should be adjusted to 100%.
- -
Volume control, pressure-limited mode is preferable and PEEP should be kept at the same level during the procedure. Adjustments can be made dynamically, with a prior assessment of the anticipated risks (e.g., lung derecruitment and desaturation, arrhythmias, pneumothorax).
- -
To avoid aerosol dispersion, a simple and appropriate maneuver consists of clamping the ventilation circuit just before introduction of bronchoscope, repeating the same step just before withdrawal.
- -
Bronchoscope removal and reinsertion should be avoided during the procedure.
- -
In hypoxemic patients, if bronchoalveolar lavage is needed for diagnostic purposes, the volume used should be reduced to a minimum. If a SARS-Cov-2 diagnosis is needed, a minimum of 2−3 mL of recovered lavage is enough.26
Rigid bronchoscopy is used for diagnostic and therapeutic purposes, in procedures where flexible bronchoscopy would be deemed difficult or even impossible, like obtaining larger samples of endobronchial lesions, foreign body removal, management of central airway obstruction (including ablative techniques, like electrocautery, argon plasma coagulation, laser, cryotherapy, among others, and placement of airway stents) or massive hemoptysis.36
There are different ventilation strategies used during rigid bronchoscopy, although manual jet ventilation and high frequency jet ventilation are much the most frequent.37 Common to these two techniques is the fact that the proximal end of the bronchoscope is open to allow the passage of instruments, thus ventilation is achieved providing 100% oxygen under high pressure (usually 50 psi) through an open system.39,40 The use of these ventilation techniques means that aerosols are released into the room, making it a high-risk procedure.
In patients with suspected or confirmed COVID-19 diagnosis, rigid bronchoscopy should be avoided, except for urgent cases (Table 1). Clinical scenarios are mostly therapeutic, like acute foreign body aspiration, massive hemoptysis (when there is no place for embolization), severe symptomatic central airway obstruction (either benign or malignant) and migrated stents. In a clinically stable patient, upon suspicion of foreign body aspiration, one should consider non-contrast computerized tomography (CT) to confirm the presence of a foreign body before rigid bronchoscopy, to avoid unnecessary exams.38,39
In some centers, the rigid scope is used to perform other techniques, like Endobronchial Ultrasound-Transbronchial Needle Aspiration (EBUS-TBNA) or transbronchial cryobiopsy; this provides comfort to the operator and safety in case of major bleeding. The authors recommend performing these diagnostic procedures through cuffed endotracheal tube to minimize the risk of exposure. However, the operator must be ready to convert to rigid bronchoscopy, if necessary.
In a patient undergoing rigid bronchoscopy, it is recommended that:
- -
Rigid bronchoscopy should always be performed in a negative pressure room.
- -
Controlled ventilation is preferred, with the rigid bronchoscope used like an endotracheal tube.
- -
Air leaks should be reduced using rubber caps on the ports of the rigid scope, as well as using a plastic covering (Fig. 2F) or filling the mouth with gauze.40 While this strategy is more appealing to minimize aerosol spread, the operator may find it challenging to handle instruments through the working channel.
Pleural effusion does not appear to be a prominent feature of COVID-19. It occurs in 5.3–5.8% of patients, according to two recent meta-analyses.41,42 There have been occasional reports of bilateral effusion that resolved spontaneously.43 As bacterial superinfection is common in severe patients, they can also develop complicated effusions or empyema, requiring targeted treatment. There have been a few anecdotal reports of spontaneous pneumothorax and pneumomediastinum in severe COVID-19 pneumonia, requiring drainage.44,45 This may be more frequent in critical patients on invasive ventilation, which can lead to bronchopleural fistulae.46 It is, therefore, plausible that pleural drainage may be necessary in some COVID-19 patients, either in the ICU or in the ward, and indications for drainage do not differ from the standard clinical guidelines. However, as with any invasive procedure in confirmed COVID-19 patients, all precautions regarding the full use of protective equipment should be taken. The procedure must be performed by trained and dedicated staff to reduce its duration and to minimize the risk of complications. In other situations, the use of ultrasound may be very helpful with COVID-19 patients.47 Besides its wide availability, safety and low cost, it is easy to use at the bedside and allows medical staff to detect small pleural effusions and to guide pleural fluid collection and drainage, if needed. On the other hand, even patients without suspected COVID-19 can have assymptomatic infection; so, any procedure should be considered as a possible COVID-19 case and precautions should be taken. Indeed, although some procedures may be postponed, in many situations they should not be deferred, especially in suspected or confirmed cancer patients. It is crucial that cancer patients do not experience delays in diagnostic or therapeutic procedures due to the present contingency.48
Few societies have published guidelines addressing pleural procedures during COVID-19 pandemic. The British Thoracic Society has issued guidance on pleural services provision,49 mainly to minimize hospital visits and admissions and to ensure both patient and staff safety. Nonetheless, although we recognize lack of published evidence supporting these reccomendations, this document will adopt some of them.
First of all, despite pleural procedures not being listed as Aerosol Generating Procedure (AGP) in the CDC updated reccomendations,50 they should be considered so and Level 2 PPE should be worn, as described above. Other societies have considered potential AGP as any procedure “likely to induce coughing, that should be performed cautiously and avoided if possible”.51 This is particularly relevant to open procedures, such as thoracoscopy and indwelling pleural catheter insertion, and in case of pneumothorax with suspected bronchial-pleural fistula. Thus, we recommend taking the following precautions:
Pleural effusion- -
On suspicion of malignant effusion, diagnostic pleural fluid aspiration should be performed, especially if the patient is a candidate for systemic therapy or if the effusion is symptomatic (Table 1).
- -
Consider placement of indwelling pleural catheters for recurrent malignant pleural effusions, avoiding repeated hospital drainage or admission.
- -
On suspicion of pleural infection, pleural fluid aspiration and analysis should be performed.
- -
In case of benign pleural effusions, the risk-benefit should be discussed with the attending physician; procedures can be postponed if the patient is not symptomatic.
- -
Thoracoscopy is not recommended as a principle; however, in the absence of alternative options, its risk-benefit must be assessed.
- -
Spontaneous primary pneumothorax can be managed in outpatient care if the risk assessment allows and if there is local team experience; needle aspiration and discharge should be considered if the patient is minimally symptomatic.
- -
When pleural drainage placement is necessary, if there is local expertise and the patient is at low risk, the use of pleural vent systems should be considered, thus allowing the patient to be managed at home.
- -
Extra care must be taken when placing the chest tubes, in order to avoid open communication with the pleural space and the potentialemission of droplets and aerossols.
- -
When chest tubes are placed in ventilated patients, consideration should be given to clamping the ventilator circuit before assessing the pleural cavity, so that positive pressure spreading of pleural air or fluid can be prevented.
- -
Whenever possible, the use of non-wired pleural drainage should be considered; it can be connected to the drainage system before insertion into the pleural cavity (closed circuit).
- -
When pleuroscopy is required, the use of one way valve trocars should be preferred to assess the pleural cavity and properly seal the entrance port of the pleuroscope.
- -
In case of prolonged air-leaks, the use of wall suction should be weighted to create a closed system.
As with other societal consensus papers, this document was developed by a restricted panel of experts from the Portuguese Society of Pulmonology; individual clinical judgment and local resources may lead to alternative perspectives. This guidance was based on the current knowledge of COVID-19, but, as new data appears, this statement should be revised in the future to accommodate updated recommendations. At present, one of the controversial assumptions is that, every patient, even if asymptomatic, should be assumed as potentially infected with SARS-CoV-2. Therefore, it is mandatory that contact precautions and proper training on donning and doffing of PPE be provided to all HCWs involved in IP. Another key element is to plan in advance and keep each IP Unit well-organized (Table 4). Although the reduction in the number of elective procedures represents one of the central strategies to improve safety, it is crucial that patients do not suffer unnecessary delays in diagnostic or therapeutic procedures due to the current contingency. Taken together, our ultimate intention is to bring full attention to this and future outbreaks or other emerging medical situations.
Conflicts of interestThe authors have no conflicts of interest to declare.