Sarcoidosis is a multisystem disease of unknown origin characterized by the presence of noncaseating epithelioid cell granulomas.
When these granulomas develop in the lymph nodes, and the patient does not meet criteria for the diagnosis of systemic sarcoidosis we refer to this as a sarcoid-like reaction.1
Because of the clinical implications of the diagnosis of lymph node metastasis, mediastinoscopy plays a key role in the differential diagnosis in these patients.
We conducted a retrospective chart review of all patients who had undergone mediastinoscopy in our hospital from 2010 to 2015 with a diagnostic of sarcoidosis or sarcoid-like reaction and history of solid organ malignancy.
Seventy-four mediastinoscopies were performed by three different surgeons. There were 9 cases of biopsy-proven presence of noncaseating epithelioid cell granulomas and the stain and the culture for acid-fast Bacilli and fungi were negative.
5 patients met criteria for the diagnosis of systemic sarcoidosis. 3 of the 4 patients who did not have a diagnosis of systemic sarcoidosis had a history of malignancy organ solid. We present these 3 cases in order to focus on sarcoid-like reaction in cancer patients.
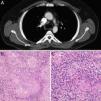
Cases reportsCase 1A 39 year-old man with a previous history of testicular seminoma, in 2012 was treated with cisplatin, bleomycin, and etoposide, after the appearance of retroperitoneal lymph node metastasis. A year later in a computed tomography (CT) control intrathoracic lymph node was revealed. It showed a 15mm premediastinal and 20mm right paratracheal lymph nodes.
In positron emission tomography combined with a computed tomography scan (PET-CT) showed premediastinal and right paratracheal lymph nodes with a maxim “standardized uptake value” (SUV) of 4.8 (Fig. 1).
There was no elevation of the tumour markers beta-human chorionic gonadotropin (beta-hCG). In view of the clinical history, we suspected a recurrence of tumour lymph nodes, and a mediastinoscoy was performed. Histopathological examination of the resected lymph nodes showed a noncaseating epithelioid cell granuloma in which no tumour cells were observed. No retroperitoneal or intrathoracic lymph nodes recurrence has been observed during follow-up.
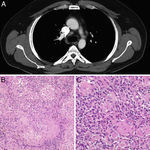
Case 2A 47 year-old man with a previous history of rectum cancer, received chemo and radiotherapy neoadjuvant, cisplatin and capecitabine. After that, low anterior rectal resection and liver metastasectomy was performed. And the treatment was completed with 12 cycles of FOLFOX and bevacizumab.
After 2 years of follow up, mediastinal lymph nodes were observed during a computed CT control. The maximum size of the lymph nodes was 17mm. Physical examination, laboratory tests, including carcinoembryonic antigen were unremarkable. In PET-CT, we observed mediastinal lymph nodes with a maxim SUV of 6.2. Endobronchial ultrasound with fine-needle aspiration (FNA) was performed but not a significant amount of histological tissue was obtained.
Mediastinoscopy and biopsy were performed, and the diagnosis was sarcoid-like reaction. No recurrence of cancer has been observed during follow-up.
Case 3A 47 year-old woman with a previous history of clear cell ovarian carcinoma, stage I, was treated with oophorectomy and adjuvant chemotherapy with cisplatin. At the time of diagnosis, mediastinal lymphadenopathy was not observed.
Six months later, CT control was performed. It revealed 24mm subcarinal and 15mm subaortic lymph nodes. A PET-CT showed subcarinal and subaortic lymph nodes with a maxim SUV of 2.53 and 3.3 respectively. Subcarinal lymph node biopsy was performed as a result of sarcoid-like reaction. She has been followed up and no recurrence of cancer has been observed.
DiscussionSarcoid-like reaction in cancer patients has been reported in 4% and 14%.2
These reactions often occur in patients with lymphoma or testicular tumour,3 and usually appear in the local lymph nodes draining. However, in our study sarcoid-like reaction is in a distant location, the mediastinum.
Sarcoid-like reaction is histologically identical to sarcoidosis, except for the presence of a population of B cells, which is not observed in sarcoidosis.4 Reactions occur because of an immune response after an aggression, whether infectious, chemical, tumour or a foreign body.
The fact that sarcoid-like reactions could appear in distant locations supports the hypothesis that it could develop from an immunological response involving T cells. The host's immune response to cancer or cancer therapy may predispose to sarcoid-like reaction, perhaps by acting as a type of auto-Kveim reagent.5
It has been theorized that certain chemotherapies may cause predisposition to the development of sarcoid-like reaction, alpha interferon, cisplatin, bleomycin or interleukin 2.1,6 In our study, all patients had been previously treated with cisplatin, so we there are reasons to suspect that this agent could develop a role in the genesis of this disease. However, chemotherapy is not used in all patients with cancer who have sarcoid-like reaction.5,6 So, the development of these diseases in these patients cannot be explained by chemotherapy alone.
Mediastinal lymphadenopathy in a patient with a previous history of malignancy requires an exhaustive diagnosis because of the potentially toxic effects of cancer therapeutics and the prognostic implications.5
PET-CT has a limited role in diagnosis. Chowdhury et al. identified a 1.1% of prevalence in sarcoid-like reaction in patients who were performing in PET-CT oncologic context.7 However, no SUV value has been determined that would enable us to discern between benign or malignant, so accurate diagnosis should always be made histologically.
Other techniques for diagnosis such as endobronchial ultrasound with fine-needle aspiration (EBUS-FNA)8 or guided biopsy CT have been reported. A study shows a sensitivity of 100%, a specificity of 93%, of EUS-FNA for the diagnosis of mediastinal sarcoidosis.9 Béchade et al. describe within a small number of patients the possibility of taking samples with the needle of 19 G to improve the histopathological examination.10
Nevertheless, mediastinoscopy takes a significant amount of histological tissue, even entire lymph nodes. It allows the diagnosis of micrometastases, which is essential for the correct diagnosis.
ConclusionSarcoid-like reaction is a disease that must be included in the differential diagnosis of patients with a history of malignancy and present with mediastinal lymph nodes.
Conflicts of interestThe authors have no conflicts of interest to declare.