The goals of the End TB strategy, which aims to achieve a 90% reduction in tuberculosis (TB) incidence and a 95% reduction in TB mortality by 2035, will not be achieved without new tools to fight TB. These include improved point of care (POC) diagnostic tests that are meant to be delivered at the most decentralised levels of care where the patients make the initial contact with the health system, as well as within the community. These tests should be able to be performed on an easily accessible sample and provide results in a timely manner, allowing a quick treatment turnaround time of a few minutes or hours (in a single clinical encounter), hence avoiding patient loss-to-follow-up. There have been exciting developments in recent years, including the WHO endorsement of Xpert MTB/RIF, Xpert MTB/RIF Ultra, loop-mediated isothermal amplification (TB-LAMP) and lateral flow lipoarabinomannan (LAM). However, these tests have limitations that must be overcome before they can be optimally applied at the POC. Furthermore, worrying short- to medium-term gaps exist in the POC diagnostic test development pipeline. Thus, not only is better implementation of existing tools and algorithms needed, but new research is required to develop new POC tests that allow the TB community to truly make an impact and find the “missed TB cases”.
Tuberculosis (TB) remains the leading infectious cause of death worldwide. The World Health Organization (WHO) estimated around 10.4 million new cases of TB in 2016 but less than two-thirds of these were diagnosed or reported to health authorities.1 The ambitious goal of the End TB strategy which aims to achieve 90% reduction in incidence and 95% reduction in mortality by 20352 will not be possible without new tools to fight TB (more effective vaccines, shorter treatment regimens, and improved diagnostic tests). Proper and rapid diagnosis is key to control TB.
“Without diagnosis, medicine is blind”3 and all other efforts directed to provide adequate and prompt treatment, and hence reduce transmission, can not be undertaken without diagnosis. Improved testing means not only developing highly sensitive and specific assays to diagnose TB and drug resistance but also tests that are affordable, rapid, and have the capacity to be deployed at the most decentralised level (point of care, POC) by health care workers with minimal training. Importantly, it is critical for programmes to view the diagnostic process holistically: for example, POC diagnosis can be improved by strengthening infrastructure at primary care, which is poor in most high burden countries (e.g., through the provision of stable electricity), without necessarily having a new testing technology.4
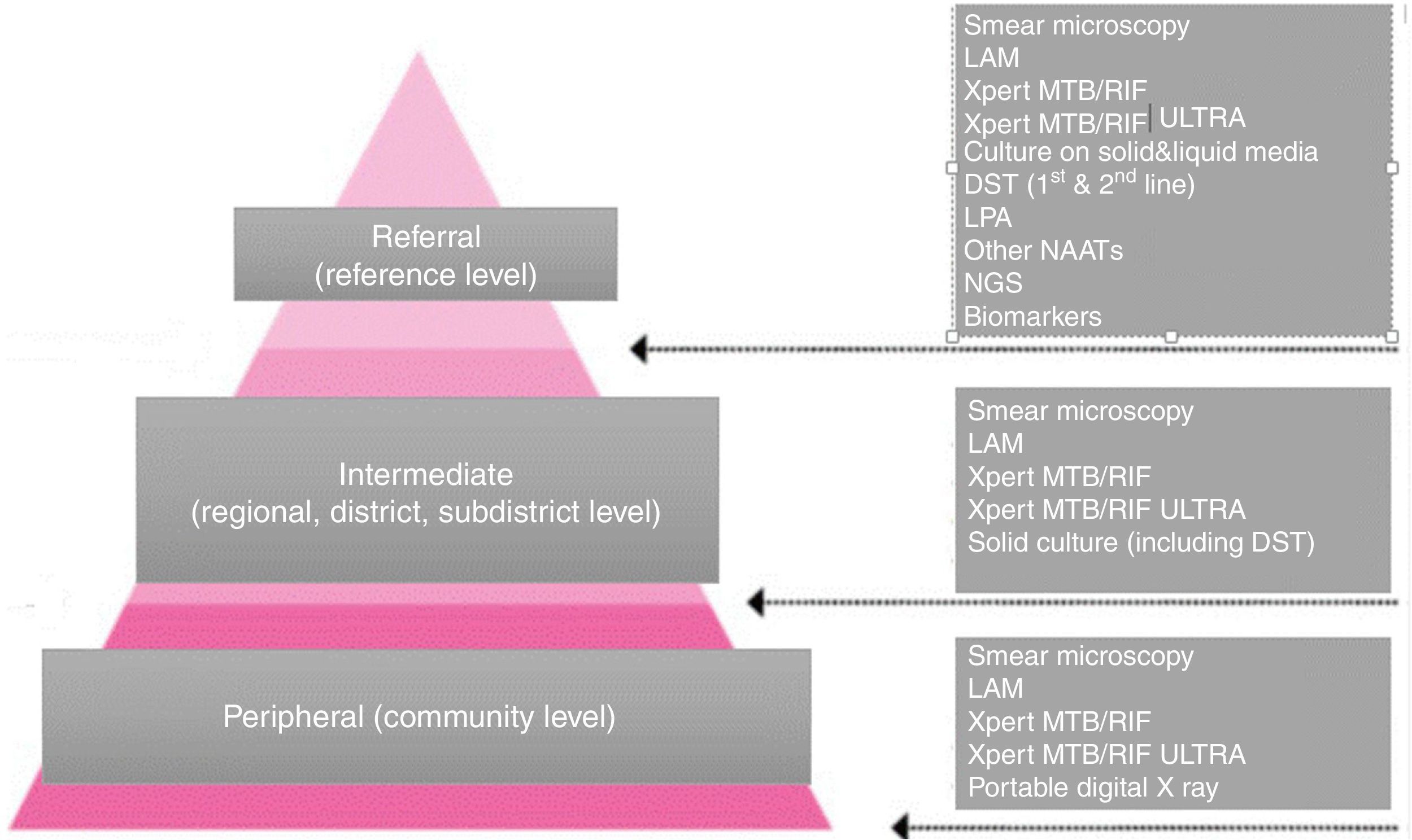
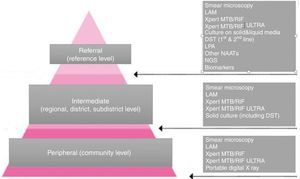
Nonetheless, in the last decade we have witnessed formidable progress in the field of TB diagnostics. Several assays, such as Xpert MTB/RIF (Xpert), Xpert MTB/RIF Ultra (Ultra), urine lateral flow lipoarabinomannan (LF-LAM) or loop-mediated isothermal amplification (TB-LAMP) have been WHO endorsed are being rolled out progressively.5–8 These “approved” tests are meant to be used at different levels of care and with different advantages and limitations (Fig. 1).
The three tiers of the network of TB laboratories and the responsibilities and the tests offered at each level.
This review aims to provide a snapshot of current assays thought to be the most useful at POC, Many of these tests do not meet the ideal characteristics of a POC test but may still be useful. We will also discuss novel future assays with POC potential.
Point of care vs centralised testingAlthough improved tests doable at health facilities with basic laboratory infrastructure are needed, there a higher urgency for tests deployable at rural TB facilities at community level. Detecting cases in these decentralised settings, often coinciding with areas with poorer health care access, is critical.
Most patients do not start treatment the day of specimen provision. There is a reasonable consensus among the research community that TB-POC tests must be deployable at the most decentralised levels of care where the patients make the initial contact with the health system, as well as in the community itself. In addition, POC tests need to lead to a rapid change in patient management (if appropriate).9 Thus, a POC should be able to be performed in an easily accessible sample and provide results in a timely manner, allowing treatment times of hours and hence avoiding patient loss-to-follow-up. We therefore considered only tests with the potential to meet these criteria, either in rural settings or well-resourced urban clinics.
Point of care test target product profileThe WHO released a series of high priority target product profiles (TPPs) for TB diagnosis at POC: (1) a non-sputum-based test capable of detecting all forms of TB by identifying characteristic biomarkers or biosignatures, (2) a triage test that can be used by first-contact health-care providers to identify those who need further testing, (3) a sputum-based test to replace smear microscopy for detecting pulmonary TB.10
The POC biomarker test (for non sputum samples) should enable the diagnosis of both pulmonary and extra pulmonary tuberculosis, paediatric TB or at early stages of the disease. It would need to be at least as sensitive as other POC tests in sputum (Xpert), portable and. The triage test would be applied to high risk patients, most of whom would not have TB and would have minimal symptoms. Thus, the test needs to be simple, low cost and highly sensitive. It is likely that this triage test would be performed in the simplest available sample, such as breath, blood or urine. The sputum replacement POC assay needs to be at least as sensitive as Xpert, robust and with a fast turnaround time without the drawbacks of Xpert (i.e., able to be used without power needs or temperature control). It is unfeasible, at least in the short term, that a single test can include all these characteristics (Table 1).
TPP of an ideal POC test for diagnosis tuberculosis.10
| Ideal characteristics of a POC assay for diagnosing Tuberculosis |
|---|
| • It can detect pulmonary or extrapulmonary tuberculosis (TB) |
| • It can be used in children and adults, HIV-positive or HIV-negative TB presumptive cases |
| • It can be used in several easily accessible body samples |
| • Used by health care workers with minimal training |
| • Used in peripheral health facilities or community |
| • It can be operated in broad ranges of temperature and humidity |
| • Delivers results in less than 20min |
| • No or minimal maintenance required |
| • Cheap (less than 4 US dollars per test) |
| • High sensitivity |
| Sensitivity similar to that of Xpert MTB/RIF for pulmonary and extrapulmonary TB |
| If intended to use as a triage test (95% compared to culture) |
| • High specificity |
| Specificity similar to Xpert MTB/RIF for pulmonary and extrapulmonary TB |
| If intended to be use as a triage test (80% compared to culture) |
Smear microscopy consists of examining specimen under a microscope to detect acid fast bacilli after staining with Ziehl–Neelsen or a Auramine.11
Sputum smear microscopy is one of the most effective tools for identifying people with infectious tuberculosis. Smear-positive patients are up to 10 times more infectious than smear-negative patients.12 The threshold of detection of AFB in sputum is 104–105CFU/ml. It is still the primary method for diagnosis of TB in low and middle income countries (LMIC).11,13 In LMIC is the only cost-effective tool for diagnosing infectious patients, monitor their progress in treatment14 and confirm cure.
Technically, smear microscopy is inexpensive, easy to perform and highly specific in areas with high prevalence. However, sensitivity values are low. Maximum sensitivity has been found to be up to 60% under optimal conditions when compared with that of cultures.15
Compared to bright-field and fluorescence microscopy, the Ziehl–Neelsen technique is easier to learn. In contrast, light emitting diode (LED) microscopy is 10% more sensitive than conventional microscopy and 98% specific11 and is considerably cheaper.16 In 2011, WHO released a new policy on LED Fluorescent Microscopy for TB.17
Despite microscopy's advantages of being cheap and rapid, results are, however, seldom on the same day.
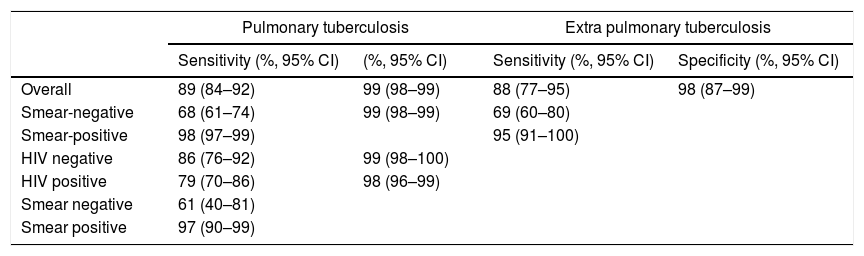
Xpert MTB/RIFXpert is a real-time quantitative PCR assay for Mycobacterium tuberculosis-complex DNA.18 It amplifies part of the rpoB gene that contains mutations that cause rifampicin-resistance (a critical first-line drug). Hence Xpert can detect TB and resistance simultaneously. Xpert has shown high sensitivity and specificity to diagnose pulmonary tuberculosis and extrapulmonary tuberculosis (Table 2).19–21 Introduced in 2011, Xpert is WHO-approved as a frontline test for pulmonary, extrapulmonary, and paediatric TB,6 undergone widespread global scale-up (∼25 million cartridges and 25,000 modules procured by end of 2016),22 revolutionised the diagnosis of TB (75% of smear-negative pulmonary TB cases can now be bacteriologically-detected within 2h), and paved the way for universal drug susceptibility testing.19,23
Xpert MTB/RIF sensitivity and specificity for pulmonary or extrapulmonary tuberculosis.19,20
| Pulmonary tuberculosis | Extra pulmonary tuberculosis | |||
|---|---|---|---|---|
| Sensitivity (%, 95% CI) | (%, 95% CI) | Sensitivity (%, 95% CI) | Specificity (%, 95% CI) | |
| Overall | 89 (84–92) | 99 (98–99) | 88 (77–95) | 98 (87–99) |
| Smear-negative | 68 (61–74) | 99 (98–99) | 69 (60–80) | |
| Smear-positive | 98 (97–99) | 95 (91–100) | ||
| HIV negative | 86 (76–92) | 99 (98–100) | ||
| HIV positive | 79 (70–86) | 98 (96–99) | ||
| Smear negative | 61 (40–81) | |||
| Smear positive | 97 (90–99) | |||
Studies have demonstrated Xpert to have POC feasibility in well-resourced clinics (largely due to its semi-automated nature that requires minimally trained non-technical personnel24–27). However, the test has, due to its need for uninterrupted power, mild ambient conditions, and economies of scale (i.e., the expensive instrumentation is most cost-effective when used at volumes that exceeds rates of specimen collection at most individual primary care facilities),28 predominantly been implemented in centralised laboratories.29
The impact of the most widespread, rapid and accurate test for TB that we have (Xpert) has thus potentially been undermined by its far-patient placement. For example, studies of POC vs centralised Xpert have shown POC placement to lead to more cases initiating treatment while maintaining similar rates of diagnostic accuracy.30,31 Thus, it appears Xpert's ability to provide POC same-day diagnoses reduces pre-treatment loss-to-follow-up.
Looking beyond Xpert, it is important to ask how can test developers and researchers improve the impact of future POC tests? How can promising research and development activity be capitalised upon, whilst being cognisant of the resource limitations at primary care in high burden settings? What are the “lessons learnt” from Xpert?
In high burden, high HIV settings, the evidence unfortunately suggests that Xpert's impact is small, with no evidence of long-term improvements in patient outcomes like morbidity and mortality.24,32,33 This is due to high rates of existing empirical TB treatment (most patients newly-detected by Xpert may have been treated on empirical grounds anyway)34 and the still suboptimal sensitivity in special populations (HIV-positives, children, extrapulmonary TB).35–38 Another important factors are weak health systems, which mean that clinics are not able to rapidly start Xpert-positive patients on treatment (this is especially acute for rifampicin-resistant patients39), the limited affordability of Xpert,40 and challenges with weak testing infrastructure at POC.41,42 Thus, when implementing successor technologies to Xpert at POC, health providers need to adopt an holistic implementation approach that includes broad systems strengthening.
Interestingly, Xpert has two additional limitations that have dampened enthusiasm in some settings, even when results are generated near POC. These limit Xpert's utility, as they increase reliance on downstream tests like culture. For example, patients with previous TB, who are an epidemiologically-important subpopulation who re-present with symptoms, have old mycobacterial genomic DNA in their lungs that can cause false-positive results43,44 (for active TB). Thus, as diagnostic tests improve in sensitivity, specificity is likely to be compromised in patients with a history of TB unless special precautions are taken. Furthermore, in the event of rifampicin-resistance detection, many countries still require confirmatory drug susceptibility testing,45 which often requires additional specimen collection, although innovative new approaches of “hacking” the used Xpert cartridge may be useful.46
Xpert MTB/RIF UltraUltra is a successor technology to Xpert that uses the same test hardware. Ultra has a limit of detection of 16CFU/ml (compared to Xpert's 114CFU/ml Xpert), and uses the same semi-quantitative categories as Xpert (high, medium, low, very low) and a new “trace” that corresponds to the lowest bacillary burden.8 If MTB is detected, category “trace”, then no interpretation can be made regarding rifampicin resistance and results are reported as ‘MTB detected, trace, RIF indeterminate.47
Overall, sensitivity of the Xpert Ultra is 5% higher than that of Xpert (95% CI +2.7, +7.8) but specificity is 3.2% lower (−2.1, −4.7). A higher incremental sensitivity is seen among paucibacillary forms of TB disease (childhood TB, HIV-associated TB, or extrapulmonary TB (1–3)). However, specificity is lower in patients with a history of TB, which means that among these patients, Ultra results should be interpreted carefully, together with a comprehensive clinical history and physical examination.8,48
Since the end of March 2017, the WHO has recommended the replacement of Xpert by Ultra. The current WHO recommendations for the use of Xpert also apply to the use of Xpert Ultra.8
GeneXpert OMNIGiven the concerns associated with the use of continuous power and need for a lab with the traditional GeneXpert platform, Cepheid has developed a POC platform. GeneXpert Omni is a single standalone and handheld module that is capable of processing Xpert cartridges in more extreme settings (elevated temperatures and humidity) and has four hours of battery life. It is intended to allow GeneXpert implementation away from central or peripheral settings. Omni is small and portable, weighing only 1.0kg. It has a supplemental battery that gives an additional 12h Battery life. As Omni module can do a single assay every ∼110min, cost and accessibility will limit adoption in high-endemic areas. The projected release of the Omni in emerging markets is at the end of 2018, and it has not yet been endorsed by WHO22 nor is there any available evidence to support its use.
Lateral flow lypoarabinomannan commercial tests (LF-LAM)Lypoarabinomannan glycolipid is a component of the mycobacterial cell wall. In 2001 a proof of concept for an ELISA detection in urine was published49 and years later a lateral flow assay for LAM detection in urine was marketed50,51 (‘Determine TB-LAM’, Alere, USA). This rapid test can be performed at the bedside, gives a result in 25minutes and costs <3 USD.
In 2015, the WHO released policy guidance7 on LF-LAM assays, stating that the test may be used to ‘assist the diagnosis of TB in HIV positive TB presumptive patients who have a CD4 count less or equal to 100cells/μL, or who are seriously ill (present with any one of four ‘danger signs’). It may also be useful in children.52,53
LF-LAM has a pooled sensitivity of 44%, however, this varies significantly according to health settings (54% in hospitalized patients and 21% in outpatients) and CD4 count (15%, 48% and 56% with >200, <200 and <100cells/μL). WHO experts discouraged its use as a screening tool (among HIV-positive patients regardless of symptoms) because of a suboptimal specificity of 92%. This low specificity was contentious because ‘false positive’ LAM results could have been signalling true TB cases but sputum negative.54 In fact, studies that sampled more bodily compartments showed LF-LAM has a specificity of 99%.55,56
LAM positivity shows a strong direct correlation with severity57,58 and mortality.56–64 The test would target precisely HIV-infected patients at risk of longer diagnostic delays (sputum negative65–68 or unable to expectorate69) and those highly immunocompromised.62,64 In 2016 a randomised clinical trial in 10 hospitals of 4 African countries showed a 17% relative risk reduction 8 week mortality regardless of CD4 when treatment initiation was guided by the addition of LF-LAM to standard diagnostics.70 Thus, LF-LAM is the only TB diagnostic test with evidence of a mortality benefit.
To better understand LF-LAM's accuracy and mortality impact, it is important to note that evidence suggests that LAM in urine mostly reflects the presence of viable mycobacteria in the kidneys,71,72 and blood dissemination. LF-LAM positivity correlates very strongly with results from autopsies showing renal or disseminated TB,71 positive mycobacterial blood culture73 and positivity Xpert in concentrated urine.69
A study in South Africa showed that clinicians exerting a very low threshold for initiating empiric TB treatment missed cases that would otherwise have been diagnosed if LAM had helped their decisions.74 WHO classic algorithms for HIV sputum negative TB suspects and seriously ill adults benefit from including LAM.59,74
Nonetheless, the HIV/TB research community continues to show that LAM in HIV-positive is useful in patients beyond those described in the WHO endorsement56,58,62,68,73–78 (or even as a prognostic52,64 or treatment response monitoring tool63). This may be due to the increasing availability of this true POC test (rapid, patient-side, low cost), a better understanding of its meaning, and the efforts driven by earlier/rapid antiretroviral treatment initiation for HIV, which can only be safe if rapid TB diagnosis is possible. Nonetheless, despite being the only new TB test with evidence for a mortality benefit, there has not been a large scale adoption of LAM. Further action and advocacy is needed to include this test within TB diagnostic screening algorithms of severely ill HIV patients.
Portable digital chest X-ray (CXR)CXR is unquestionably a very sensitive test. A recent prevalence survey in Kenya showed 92% sensitivity for TB in HIV-positive and 100% in HIV negative, but also low specificity (73%) with variations depending on the criteria used to consider “positive” certain abnormalities.79 CXR is especially useful for screening high-risk populations, for general populations in prevalence surveys, or diagnosis of smear negative TB (HIV positive TB cases or paediatric TB). The test is more sensitive (and no less specific) than asking for symptoms, except when we consider ‘any symptom’, in which case the sensitivity is similar. When used in decision trees or algorithms, CXR should be placed at the start or in the early stages, rather than at the end (confirmatory).80 Unfortunately, the latter is general practice, but only because of operational and logistical constraints that limit its availability to central facilities.
This is changing today. There is recognition that CXR is a good tool in active case finding strategies, filling the gap that symptom-based screening (SBS) leaves: it finds cases that the latter misses (specially among HIV-positive and other vulnerable populations). Combining both, and adding a rapid molecular test for laboratory confirmation of the disease is an ideal and cost-effective strategy (CXR serving as a triage to reduce the number of Xpert tests).81 The development of digital and portable X-ray systems and, importantly, automated software that obviates the need for an experienced reader, allows to CXR to be considered POC test.82 A simple van can carry an X-ray device and GeneXpert to any location.
Mounting evidence shows the success of these so-called mobile units in three scenarios (Fig. 1):
- (1)
Detecting TB in hard to reach risk populations in low incidence/high income countries.83,84 A recent systematic review85 showed this is a highly cost-effective strategy.
- (2)
High incidence/medium income countries such as India,86,87 Myanmar88 or Philippines89 are increasingly using mobile clinics to bring GeneXpert and Xray to peripheral/rural facilities where TB suspects are periodically appointed for a one-stop (same day) diagnosis.87
- (3)
Active case finding campaigns in aggregated settings (prisons, hospices).89,90
The digitalization not only allows portability but also tele-medicine91 and computer aided diagnosis (CAD). Their specificity, which increases by adding clinical information,92 is already coming close to human interpretation93 (except by experienced radiologists)81 making them a very attractive option in low-resourced settings and remote areas.94
Novel/future assays with potential for POC for TB diagnosisNAATs and next generation sequencing for TB diagnosis (and detection of drug resistance)Despite the recent advances in automated PCR technology,95 what are the next PCR/NAAT technological enhancements necessary for a same-day point of care test? The combination of decentralised health infrastructure and new technology improved ATT initiation from 64 to 18 days.96 There are numerous other potential improvements that could give clinicians the ability to diagnose TB and drug resistance on the same day as seeing the patient.
In addition to Xpert, there are other real-time PCR assays including, but not limited to, Artus MTB (Qiagen, Germany), Molbio (Bangalore, India), Anyplex II MTB/MDR (Seegene, Korea) and Abbott RealTime MTB. While these assays are less automated than Xpert, the isolated DNA can be paired with other assays, such as paper-based drug detection via line probe assays. The Abbott assay added additional Mtb gene targets to improve sensitivity.97 Most of these assays have a relatively small amount of evidence to support their use, require a thermocycler as well DNA isolation and therefore are appropriate in central and some peripheral laboratory settings. To push PCR towards peripheral and community-based testing separate advances are necessary.
Potential future POC diagnostic PCR/advances could come from (1) isothermal technology; (2) paper-based innovations; (3) quantitative DNA±RNA assays; and (4) improvements in DNA isolation from non-invasive, non-sputum specimens such as urine, stool or dry blood spot. Loop-mediated isothermal amplification (LAMP) has been used extensively, with multiple meta-analysis describing sensitivity and specificity of ∼89–93% and 94–95% respectively.98–100 In contrast to RT-PCR using a forward and reverse primer, LAMP uses four to 6 primers, does not require an expensive thermocycler and has similar limits of detection (LOD). As it does not need thermocycler or fluorescence detection, LAMP technology has the potential to decrease test cost and time to diagnosis in peripheral labs or community settings.
Similarly, a paper-based assay like the lateral flow assays (LFAs) used for at home pregnancy and HIV testing shows promise for improving TB diagnostics. Proof of concept for nucleic acid LFAs have been successful detecting Mtb at 104 copies of genomic DNA as well as katG and rpoB genotypic resistance.101 The EasyNAT (Ustar, China) incorporates a syringe-based minimally laborious DNA extraction step with isothermal amplification (no expensive thermocycler required) followed by a simple Lateral flow detection method (LF).102 While advances in portability, simplicity and eliminating the need for a thermocycler, improvements in limit of detection (LOD), currently >104CFU/ml, will be needed for ideal sensitivity.103
The majority of PCR/s are designed for evaluating sputum specimens, however large proportions of community and peripheral health centres are not capacitated to induce sputum and certain populations (PLHIV, infants, elderly) are unable to expectorate. Modifications of existing tests to use minimally-invasive specimen types such as urine, stool or string test offer the promise of increasing diagnostic yield, genotypic drug resistance detection and treatment monitoring in patient populations more likely to suffer poor outcomes.104–108
NAATs and next generation sequencing for detection of drug resistanceIn combating the rising number of cases of drug resistant TB, the ability of Xpert to quickly detect rpoB genotypic resistance is improving time to starting appropriate therapy. In order to prescribe the recently WHO-recommended shorter regimen, clinicians should be sure that RR/MDR-TB patients are not resistant to these drugs, thus samples need to be referred to an intermediate level laboratory for rapid testing.109–116 As the open and manual format of LPAs and the technical requirements of culture, preclude their use as POC tests,117 it is critical that POC clinics have rapid referral mechanisms in place for specimens for further DST. The advent of the Xpert XDR cartridge might help further DST to be done more widely.109
Next-generation sequencing (NGS) is the next advance in rapid detection of genotypic resistance with initial studies suggesting the ability to identify resistance earlier and potentially with improved cost effectiveness compared to phenotypic drug susceptibility testing (DST). Preliminary NGS studies detect genotypic resistance with 97–100% concordance compared phenotypic DST.118–120 Similar to the line probe assays, it is quite feasible for NGS to be performed directly on smear-positive clinical sputum samples119–121 with INH and RIF resistance sensitivities and specificities of 95–97% and 98–100%, respectively. Compared to phenotypic DST, the potential improvements in times to drug resistance detection ands cost provide cautious optimism.
To become a point of care, or even near point of care in very well-resourced setting, sequencers will need to be portable (the MinION is an early example of this) and complex bioinformatic algorithms will continue to advance in efficacy and timeliness. When implementing NGS directly from sputum, urine or stool, improvements in bacillary enrichment will decrease background noise from other microbes present in sputum and non-sputum based samples. Additional potential benefits from NGS include enhanced contract tracing, detection of subpopulations (heteroresistance) and identification of pathogen virulence factors (further in-depth reviews of the hopes and hindrances of NGS for TB diagnostics are available).122–124
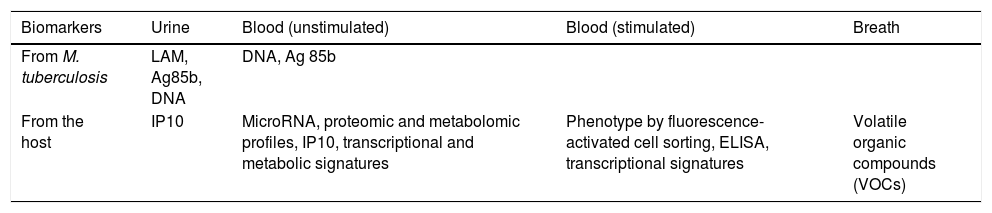
Biomarkers (host and pathogen)A biomarker is a substance, structure or process that can predict the incidence or outcome of a disease.125 To be used at POC, biomarkers need to perform in settings with limited laboratory facilities and ideally be low cost and easy to use.126 In the last decade, multiple biomarkers with diagnostic potential which could be developed as POC assays in the future127 have been identified. However very few are currently available. A recent review of 399 non-DNA potential biomarkers suitable for the POC concluded that only 12 were validated prospectively and only one had reached WHO endorsement for patients with advanced HIV (lipoarabinomannan, LAM, already discussed).128 Here we describe examples of some of these other biomarkers with potential use as POC tests (Table 3):
Host and pathogen biomarkers with potential for POC diagnosis of Tuberculosis (Modified from Goletti, 2016).
| Biomarkers | Urine | Blood (unstimulated) | Blood (stimulated) | Breath |
|---|---|---|---|---|
| From M. tuberculosis | LAM, Ag85b, DNA | DNA, Ag 85b | ||
| From the host | IP10 | MicroRNA, proteomic and metabolomic profiles, IP10, transcriptional and metabolic signatures | Phenotype by fluorescence-activated cell sorting, ELISA, transcriptional signatures | Volatile organic compounds (VOCs) |
Mtb Ag85: The Mtb Ag85 complex is a family of three proteins (Ag85A, Ag85B, and Ag85C) with enzymatic mycolyl transferase activity involved in the coupling of mycolic acids to the arabinogalactan of the cell wall and in the biogenesis of the cord factor. Although initially promising, its detection in blood and urine shows highly variable performance129,130 and is not validated as a POC assay.
Volatile organic compounds (VOCs): Exhaled breath may contain VOCs that could be derived directly from Mtb and/or the host (e.g., products of oxidative stress). The detection of VOCs is difficult because they are excreted in picomolar concentrations and no devices are available for the POC. Several e-nose prototypes (i.e., eNosi Aeonose), point-of-care electronic nose device to diagnose TB through exhaled breath are in development, typically reporting sensitivities of 80%-88% and specificities of 70–90%.131,132
Acute phase proteins: recent studies have revisited the use of acute phase proteins such as C-reactive protein (CRP) and Alpha-1-acid glycoprotein as triage tests for TB.133 Systematic reviews have described that CRP has a sensitivity of 90–95% with a specificity that ranges from 50% to 70% depending on the setting. A prospective study in HIV positive individuals recently reported 89% sensitivity results against culture, and similar sensitivity as the WHO symptom-based screening when compared to Xpert MTB/RIF.133 CRP tests are already available in a portable and designed lock format and hence this is a potential marker which could be used for screening purposes within active case finding strategies.
A recent proteomic analysis of 1,470 samples from patients described six host response markers including tryptophanyl-TRNA synthetase, kallistatin, complement C9, gelsolin, testican-2, and aldolase C which had performed well in a training set to distinguish TB and non-TB samples. Differential expression was also high for many novel proteins not previously associated with TB such as SAA, secreted phospholipase A2 (NPS-PLA2), and carbonic anhydrase 6 (CA6).134
A pleural fluid adenosine deaminase (ADA) >35U/L has been reported to have a sensitivity of 93%, with 90% specificity for the diagnosis of TB in lymphocytic exudates. A high ADA level is also detected in neutrophilic TB effusions. However, extremely high ADA activity should raise suspicion of empyema or lymphoma.135 As ADA levels decrease with treatment, this is a potential biomarker of disease recovery.136,137 ADA exceeds Xpert's sensitivity for pleural and pericardial TB,138,139 for which unstimulated interferon-gamma is another promising biomarker.
The combined use of ascitic fluid ADA and serum CA-125 in 30 patients reported a sensitivity, specificity of ADA of 87%.
Several other human biomarkers have decreased concentrations after TB treatment initiation140 with significant correlations between CRP, serum amyloid protein A (SAA), vascular endothelial growth factor A (VEGF-A), soluble interleukin 2 receptor-alpha (sI2R-A)/CD40, and γ-interferon (IFN) inducible protein 10 (IP10) and delayed smear culture conversion.140–142 IP10 has been reported to be high in unstimulated plasma of children and adults with active TB.143 A combination of monocyte chemoattractant protein-1 (MCP-1)/C-C motif chemokine 2 (CCL2), IP-10, sIL-2Rα, SAA, CRP and smear microscopy smear grade could distinguish fast from slow responders and were predictive of delayed smear culture conversion with 86% and 83% sensitivity and specificity, respectively. Thus, some of these biomarkers could play a role in the identification of patients put under treatment without having TB (among those clinically diagnosed), or those with drug resistant TB put under drug susceptible TB combinations.144,145
The neutrophil driven interferon (IFN)-inducible gene profile (Type 2 [IFNγ) and Type I (IFNαβ) IFN signalling] has a TB signature detectable in the peripheral blood and its levels decrease with effective treatment.146
The combined use of ascitic fluid ADA and serum CA-125 in 30 patients reported a sensitivity, specificity of ADA of 87% and 83% respectively, whereas CA-125 had 83% sensitivity and 50% specificity.147
Although many of these markers are promising, none of these biomarkers, except LAM and acute phase proteins, are available in POC format and there are no prototype diagnostics at this stage.
Other novel assaysPoint-of-care diagnostic development should combine the most innovative technologies with effective early detection and affordable costs.
For some time, considerable attention has been devoted in the bioelectronic field. Biosensors are analytical devices that combine a biological sensing element with a physicochemical transducer. The importance of biosensors results from their high analytical specificity and sensitivity, which allows the detection of a broad spectrum of analysts in complex sample matrices.148 Importantly, a biosensor can only (in ideal situations) perform as well as the association of the biomarker it is designed to detect with the condition of interest.
Also, other properties, such as being easy-to-operate, economical, portable and real-time results provider, identify them as promising future diagnostic methods. Nucleic acid or antibody based biosensors have been built to detect different molecular targets. There are several promising assays aimed at finding whole bacteria or defining specific MTBC antigens: an amperometric immunosensor to detect MTB cells in sputum, captured on a microtip surface and detected by electric current149 or an immuno-sensor development of ESAT-6, based on a voltammetry method,150 are only some of them.
The combination of nanotechnology and biosensing technology has great potential in the medical diagnostics field.148 A magnetoresistive biosensor to detect BCG bacteria,151 a lab-on-chip (LOC) platform which can perform label-free and rapid single-cell capture152 or a colorimetric sensing strategy employing gold nanoparticles153 are very promising assays.
Other tantalizing developments with potential for POC could be transcriptional signatures of host blood. An interferon-inducible neutrophil-driven blood transcriptional signature distinguished pulmonary tuberculosis from other respiratory or granulomatous diseases or cancers.154 Other relevant study showed that host mRNA signature from host blood can differentiate tuberculosis from other diseases in several cohorts of African children, regardless of HIV infection.155 However, the equipment and complexity involved in the analysis of transcriptional signatures preclude their use in POC for now.
ConclusionAlthough we are emerging from a period of unprecedented research, development, and implementation for new TB diagnostics, we have a limited number of near POC tests, and even fewer commercially-available true POC tests. None of these meet any of the WHO TPP criteria but they still are useful and, in the case of LF-LAM for example, have compelling evidence to support their implementation. Furthermore, these is a worrying gap of late stage potential POC tests for TB. Finally, not only can the simplicity of tests be improved so they are deployable at the POC, but the conditions at the POC can themselves be raised, so that more sophisticated tests are doable. This requires a holistic approach to ending TB.
Conflicts of interestGT has received consumables donations from Cepheid, Alere, and Hain LifeSciences. GT has consulted for the WHO.
The paper is part of the ERS/ALAT and the ERS/SBPT collaborative projects (ERS: European Respiratory Society; ALAT: Latino-American Society of Respiratory Medicine; SBPT: Brazilian Society of Pulmonology).