Vaccination has become one of the main public health measure for influenza control, its role in reducing the risk of developing the disease and post-infection complications1 is widely recognised. Influenza vaccination is recommended for the elderly, for individuals with chronic conditions, the pregnant and health professionals. Due to the continuous changes of the influenza virus, the seasonal flu vaccine composition is reformulated annually, to match viruses that are expected to circulate in that season. Early in the season influenza vaccine effectiveness (IVE) estimate is an important indicator of the need for further public health measures, it is critical when the IVE is low.
This study aimed to provide early 2017/2018 seasonal IVE using data from the Portuguese influenza surveillance system.
MethodsThis analysis included patients with influenza-like illness (ILI) from primary care and emergency departments swabbed for the detection of influenza using RT-PCR and reported to the National Influenza Surveillance System (NISS)2 between weeks 38/2017 to 5/2018. We used the test-negative case–control study design,3 where influenza laboratory confirmed incident ILI patients (Cases) were compared to laboratory influenza negative ILI patients (Controls). A medical doctor collected demographic and clinical data, ILI signs and symptoms, onset and swab collection dates and vaccination status during the consultation or through the clinical record. An ILI patient was considered vaccinated if the 2017/2018 influenza vaccine uptake occurred 14 days before symptom onset. IVE analysis was restricted to ILI patients with symptoms compatible with the European Union ILI definition,4 i.e., with sudden onset of at least one systemic and one respiratory symptom. Chi-square test was used to compare baseline characteristics between Cases and Controls. Crude IVE, design adjusted for calendar time, was estimated using 1-odds ratio (OR) of being vaccinated in Cases vs. Controls and was further adjusted for confounding by age group and presence of chronic condition.
ResultsA total of 732 ILI patients were reported to the NISS and approximately 74% adhered to the ILI definition (Table 1).
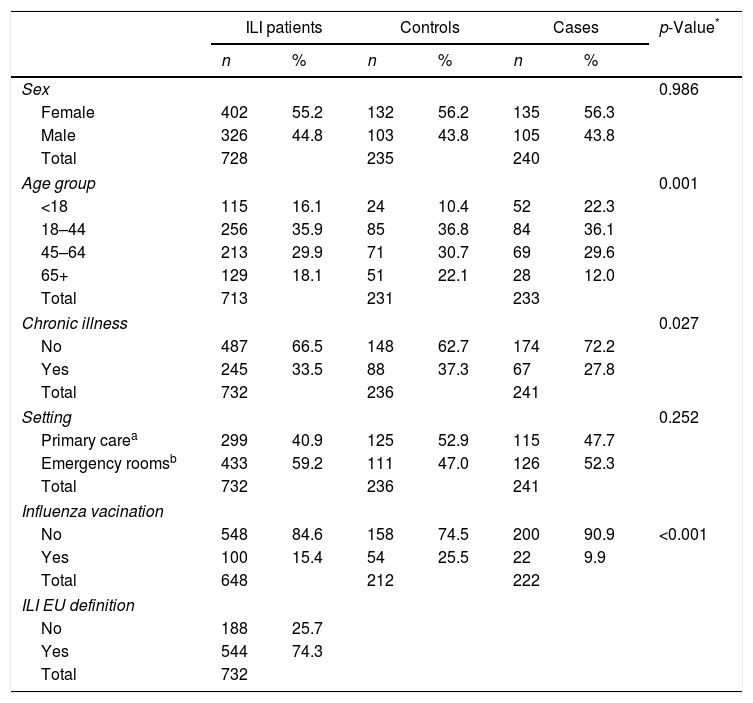
Description of notified ILI patients and comparison of Cases and Controls according to sex, age group, presence of chronic condition with indication to vaccine uptake, setting, influenza vaccine uptake and ILI clinical definition.
| ILI patients | Controls | Cases | p-Value* | ||||
|---|---|---|---|---|---|---|---|
| n | % | n | % | n | % | ||
| Sex | 0.986 | ||||||
| Female | 402 | 55.2 | 132 | 56.2 | 135 | 56.3 | |
| Male | 326 | 44.8 | 103 | 43.8 | 105 | 43.8 | |
| Total | 728 | 235 | 240 | ||||
| Age group | 0.001 | ||||||
| <18 | 115 | 16.1 | 24 | 10.4 | 52 | 22.3 | |
| 18–44 | 256 | 35.9 | 85 | 36.8 | 84 | 36.1 | |
| 45–64 | 213 | 29.9 | 71 | 30.7 | 69 | 29.6 | |
| 65+ | 129 | 18.1 | 51 | 22.1 | 28 | 12.0 | |
| Total | 713 | 231 | 233 | ||||
| Chronic illness | 0.027 | ||||||
| No | 487 | 66.5 | 148 | 62.7 | 174 | 72.2 | |
| Yes | 245 | 33.5 | 88 | 37.3 | 67 | 27.8 | |
| Total | 732 | 236 | 241 | ||||
| Setting | 0.252 | ||||||
| Primary carea | 299 | 40.9 | 125 | 52.9 | 115 | 47.7 | |
| Emergency roomsb | 433 | 59.2 | 111 | 47.0 | 126 | 52.3 | |
| Total | 732 | 236 | 241 | ||||
| Influenza vacination | |||||||
| No | 548 | 84.6 | 158 | 74.5 | 200 | 90.9 | <0.001 |
| Yes | 100 | 15.4 | 54 | 25.5 | 22 | 9.9 | |
| Total | 648 | 212 | 222 | ||||
| ILI EU definition | |||||||
| No | 188 | 25.7 | |||||
| Yes | 544 | 74.3 | |||||
| Total | 732 | ||||||
Statistical significant differences were verified in the age group distribution between Cases and Controls (Table 1). Cases presented a lower proportion of at least one chronic condition and vaccine coverage.
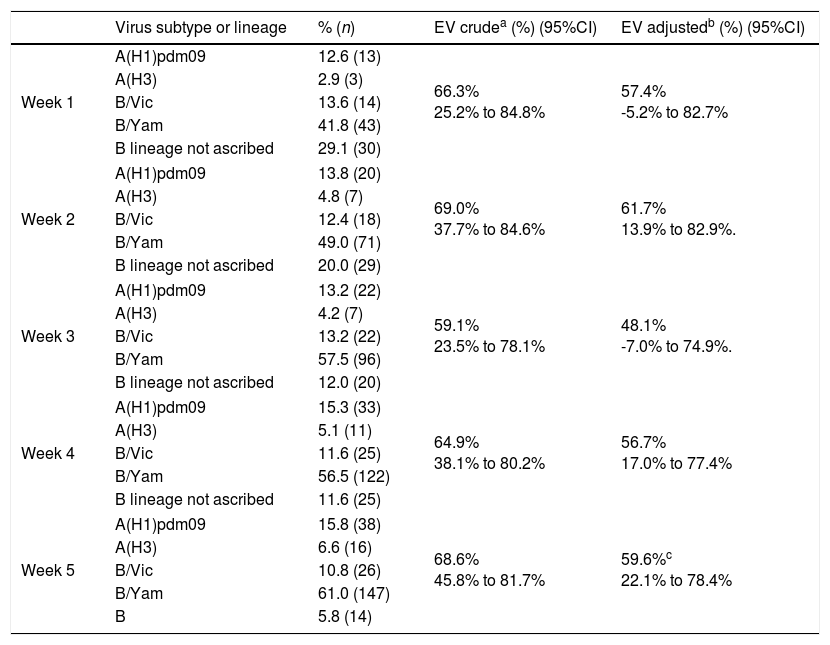
Using data updated up to week 5 (Table 2), adjusted IVE was estimated in 59.6% (95%CI: 22.1–78.4%). Considering the weekly IVE monitoring, results observed at the end of the period were very similar to the ones obtained at the beginning of the epidemic period (week 2) (even though with low precision). This result highlights the potential of having very early indicative estimates using surveillance data.
Crude and adjusted IVE against medically attended influenza, week 1 to week 5.
| Virus subtype or lineage | % (n) | EV crudea (%) (95%CI) | EV adjustedb (%) (95%CI) | |
|---|---|---|---|---|
| Week 1 | A(H1)pdm09 | 12.6 (13) | 66.3% 25.2% to 84.8% | 57.4% -5.2% to 82.7% |
| A(H3) | 2.9 (3) | |||
| B/Vic | 13.6 (14) | |||
| B/Yam | 41.8 (43) | |||
| B lineage not ascribed | 29.1 (30) | |||
| Week 2 | A(H1)pdm09 | 13.8 (20) | 69.0% 37.7% to 84.6% | 61.7% 13.9% to 82.9%. |
| A(H3) | 4.8 (7) | |||
| B/Vic | 12.4 (18) | |||
| B/Yam | 49.0 (71) | |||
| B lineage not ascribed | 20.0 (29) | |||
| Week 3 | A(H1)pdm09 | 13.2 (22) | 59.1% 23.5% to 78.1% | 48.1% -7.0% to 74.9%. |
| A(H3) | 4.2 (7) | |||
| B/Vic | 13.2 (22) | |||
| B/Yam | 57.5 (96) | |||
| B lineage not ascribed | 12.0 (20) | |||
| Week 4 | A(H1)pdm09 | 15.3 (33) | 64.9% 38.1% to 80.2% | 56.7% 17.0% to 77.4% |
| A(H3) | 5.1 (11) | |||
| B/Vic | 11.6 (25) | |||
| B/Yam | 56.5 (122) | |||
| B lineage not ascribed | 11.6 (25) | |||
| Week 5 | A(H1)pdm09 | 15.8 (38) | 68.6% 45.8% to 81.7% | 59.6%c 22.1% to 78.4% |
| A(H3) | 6.6 (16) | |||
| B/Vic | 10.8 (26) | |||
| B/Yam | 61.0 (147) | |||
| B | 5.8 (14) | |||
The 2017/2018 season in Portugal has been characterised by A(H1)pdm09, AH3 and B viruses co-circulation, with predominance of B/Yamagata lineage. Considering the composition of the seasonal trivalent vaccine, which contains B/Victoria component, this constitutes a lineage mismatch which potential affects IVE.
However, our results indicate that the 2017/2018 seasonal vaccine conferred moderate protection against medically attended influenza. Given that the majority of cases were positive for B/Yamagata (61%), this could reflect some cross-lineage protection. Similar results were already reported in Canada, where IVE against B cases was estimated in 55% (95% CI: 38–68%), also in a context of B/Yamagata virus dominance.5 The results obtained this season are also in line with meta-analysis of IVE by virus subtype.6
Our results were estimated using surveillance data, not collected from IVE estimates, with missing information on some relevant factors. For this reason, from the initial sample of ILI patients, 74% were included in the analysis. In addition, this limited the number of variables that could be included in the model as confounders, particularly in earlier weeks due to low sample size. Nevertheless, and besides this limitation, when comparing week 2 with week 5 estimates (which were adjusted for more variables), IVE point estimates remained stable.
ConclusionsIn a season with B/Yamagata dominance and with vaccine lineage mismatch, the 2017/2018 trivalent seasonal influenza vaccine conferred moderate protection against medically attended influenza.
The use of surveillance data constituted a useful tool to have early in the season IVE estimates.7 These results assist modifications to health interventions, such as using antiviral treatment in high-risk patients, reinforcement of social eviction and individual hygiene measures to reduce risk of influenza transmission, regardless of the vaccination status.
Conflicts of interestThe authors have no conflict to declare.
EuroEVA project: Verónica Gómez, Department of Epidemiology, National Health Institute Dr. Ricardo Jorge, Lisbon, Portugal. Inês Costa, Department of Infectious Diseases, National Health Institute Dr Ricardo Jorge, Lisbon, Portugal. Paula Cristóvão, Department of Infectious Diseases, National Health Institute Dr Ricardo Jorge, Lisbon, Portugal. Pedro Pechirra, Department of Infectious Diseases, National Health Institute Dr Ricardo Jorge, Lisbon, Portugal.