The diagnosis of cancer during pregnancy is rare and constitutes a clinical challenge because of treatment implications and potential risk for the pregnant woman and the fetus.1,2 The authors describe a case of advanced lung cancer diagnosed during pregnancy and its therapeutic approach.
The patient was a 36 years-old pregnant female, with previous history of lower limbs varicose veins surgery. There was no other relevant medical history or family history of oncological diseases. At 18 weeks of gestation, the patient presented to the emergency department complaining of non-productive cough, dyspnea on exertion, left posterior pleuritic chest pain and asthenia; with no fever associated. Decreased breath sounds in left hemithorax were noted. A left pleural effusion was detected on chest radiograph and a thoracentesis was performed, leading to her hospitalization at 22 weeks of gestation. Pleural fluid had characteristics of an exudate and cytology was compatible with adenocarcinoma of primary pulmonary origin [thyroid transcription factor 1 (TTF-1) positive staining], positive for echinoderm microtubule-associated protein-like 4-anaplastic lymphoma kinase (EML4-ALK) translocation. For dyspnea relief, a chest tube was inserted. No relevant abnormal findings were detected on breast and abdominal ultrasound or on abdominopelvic magnetic resonance. Thoracic computed tomography (CT) revealed a pulmonary mass in the left lower lobe, with perihilar localization and partial atelectasis of this lobe, as well as interlobular septal thickening, suggestive of lymphangitic carcinomatosis (Fig. 1). Talc pleurodesis was performed, due to fast pleural effusion recurrence, which proved to be effective. At the multidisciplinary team board meeting, it was decided to start targeted therapy with crizotinib 250mg bid, at 26 weeks of gestation. No side effects related to this therapy were detected, however, no clinical or radiological improvement was observed. Meanwhile, there was disease progression with a breast metastasis of lung adenocarcinoma. Due to worsening of general condition, it was decided to perform caesarean delivery at 30 weeks of gestation. The newborn was healthy, but adenocarcinoma cells were identified in placental pathological examination. At disease restaging (Fig. 2), skeletal scintigraphy confirmed the presence of multifocal bone metastasis and abdominal ultrasonography showed multiple hepatic metastasis and ascites, with positive cytology. On D45 crizotinib, 15th day of puerperium, due to the rapid progression, it was decided to suspend it and initiate chemotherapy with carboplatin and pemetrexed, while waiting for ceritinib approval, completing 2 cycles of chemotherapy. Subsequently, the patient initiated ceritinib, dying 15 days later. To date, the child follow-up at 29 months exhibited regular development.
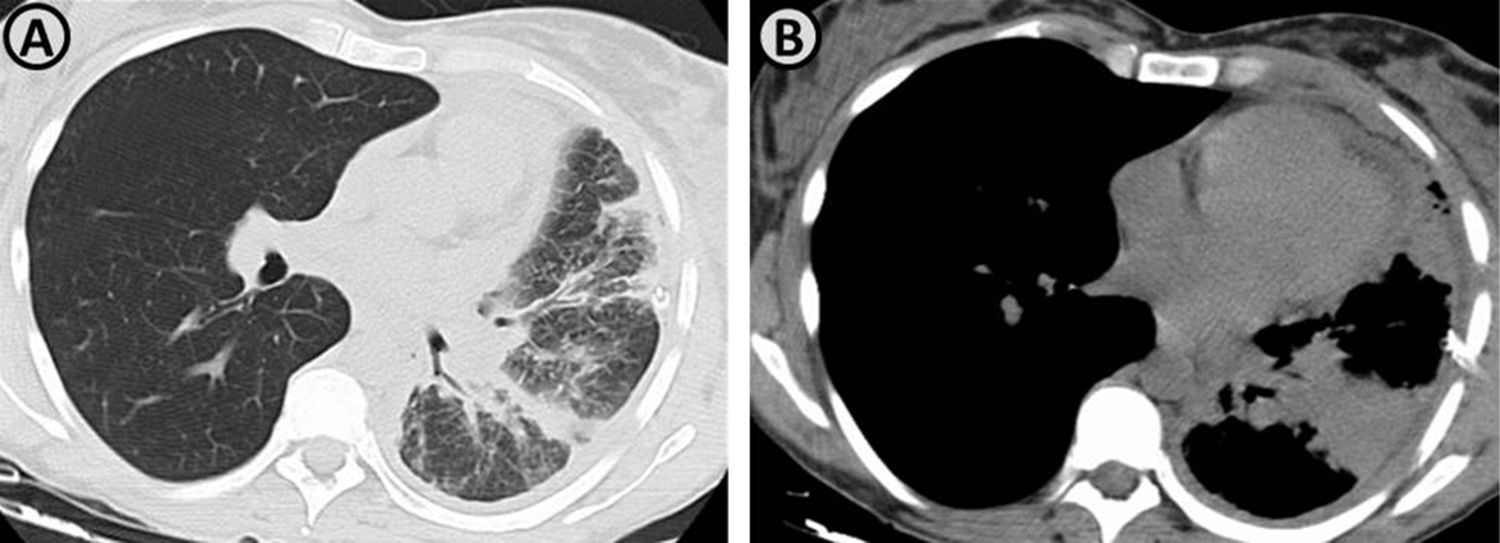
(A and B) Axial thoracic computed tomography images demonstrate the presence of a pulmonary mass in the left lower lobe, with perihilar localization, conditioning partial atelectasis of this lobe. There is also seen interlobular septal thickening, suggestive of lymphangitic carcinomatosis.
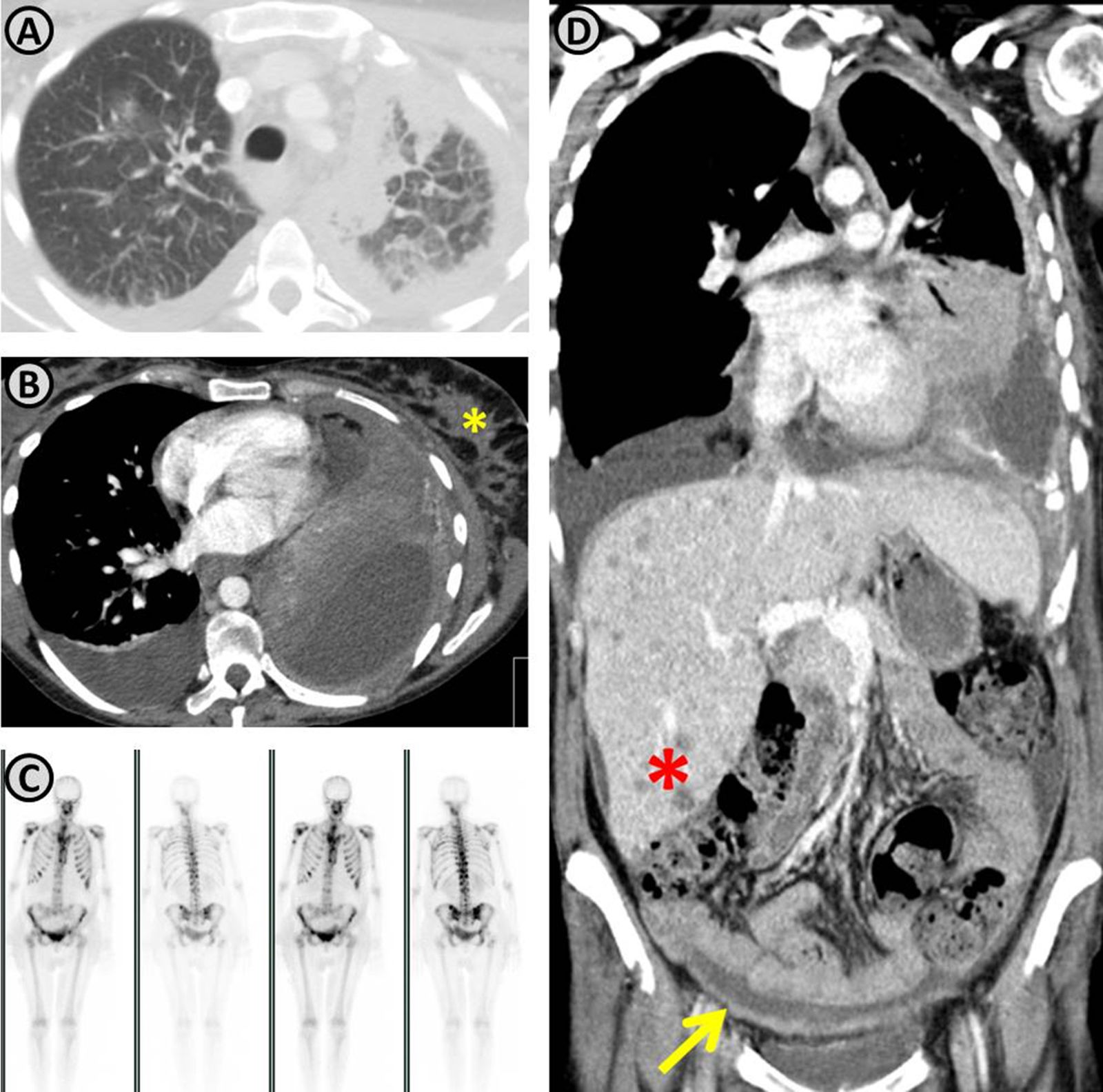
Restaging demonstrating disease progression. (A and B) Increasing of the lung mass and the extent of lymphangitic carcinomatosis; in the mediastinal images (B) a breast metastasis is observed (yellow asterisk). (C) Presence of multifocal bone metastasis. (D) Coronal reconstruction demonstrating progression of the lung mass, multiple hepatic nodules (red asterisk) suggestive to be metastatic, and peritoneal thickening with ascites (yellow arrow) compatible with peritoneal carcinomatosis.
Given the lack of safety profile studies of the different chemotherapy regimens and target therapies during pregnancy, it is essential to report clinical cases and to share evidence about the use of antineoplastic therapies during the gestation period. This is of utmost importance in an era in which the incidence of lung cancer during pregnancy is expected to increase, which might be partially related to higher mean age of pregnancy and increase in tobacco consumption by women.1–3 As far as we know this is the first case report of administration of crizotinib, an anti-neoplastic target therapy, during pregnancy. In this case, this therapy does not seem to have caused any harm to the newborn; however, a longer follow-up period will be necessary to assess the exposed child. In fact, most cases of lung cancer in pregnancy are diagnosed in an advanced stage and might progress to dyspnea which is difficult to relieve or a life threatening condition, which strongly influences the therapeutic options.1–3 It is a huge challenge to decide on the best approach and management of cancer during pregnancy, attempting to choose the optimal anti-cancer regimen to treat the pregnant women without harming the developing fetus. Most chemotherapeutic agents are considered relatively safe when administered in the second and third trimesters and are not associated with major congenital malformations, although an increase in the risk for preterm delivery and intrauterine growth restriction has been reported.2 Besides that, in many cases commencing antineoplastic therapy cannot be postponed until after delivery. Moreover, recently, there has been an increased use in targeted anti-cancer therapies, which adds new challenges to the selection of cancer treatment during pregnancy, since the safety of these agents in this context has not been established.2 However, reports on the use of epidermal growth factor receptor tyrosine kinase inhibitor (EGFR-TKI) suggest that this treatment has not apparently been associated with consequences or major side effects on children.4–6 In relation to crizotinib neither previous reports nor any experimental animal model to access teratogenicity are available. There was a recent work which assessed the extent of placental disposition of crizotinib, in which an ex vivo model was used and showed that the drug is strongly accumulated in placental tissue.7 Therefore, the report of additional clinical data on anti-neoplastic target therapy, such as crizotinib during gestation should be encouraged since the evidence for its use is scarce and limited to case reports.
FundingThe authors declare that no funding was received for this paper.
Conflicts of interestThe authors have no conflicts of interest to declare.
Address of the institution at which the work was performed: Alameda Prof. Hernâni Monteiro, 4200-319 Porto, Portugal.