In the last 20 years, 15.8% of lung transplants were performed in patients with Cystic Fibrosis (CF). These patients present the best long term outcome and survival, with particular importance in transplant function, which is one of the aspects directly implicated in survival.1
A female patient diagnosed with CF (homozygous F508del) with lung, pancreatic and sinus involvement, colonized with Pseudomonas aeruginosa (mucoid strain) before transplantation, was submitted to bilateral lung transplant when she was 32 years old.
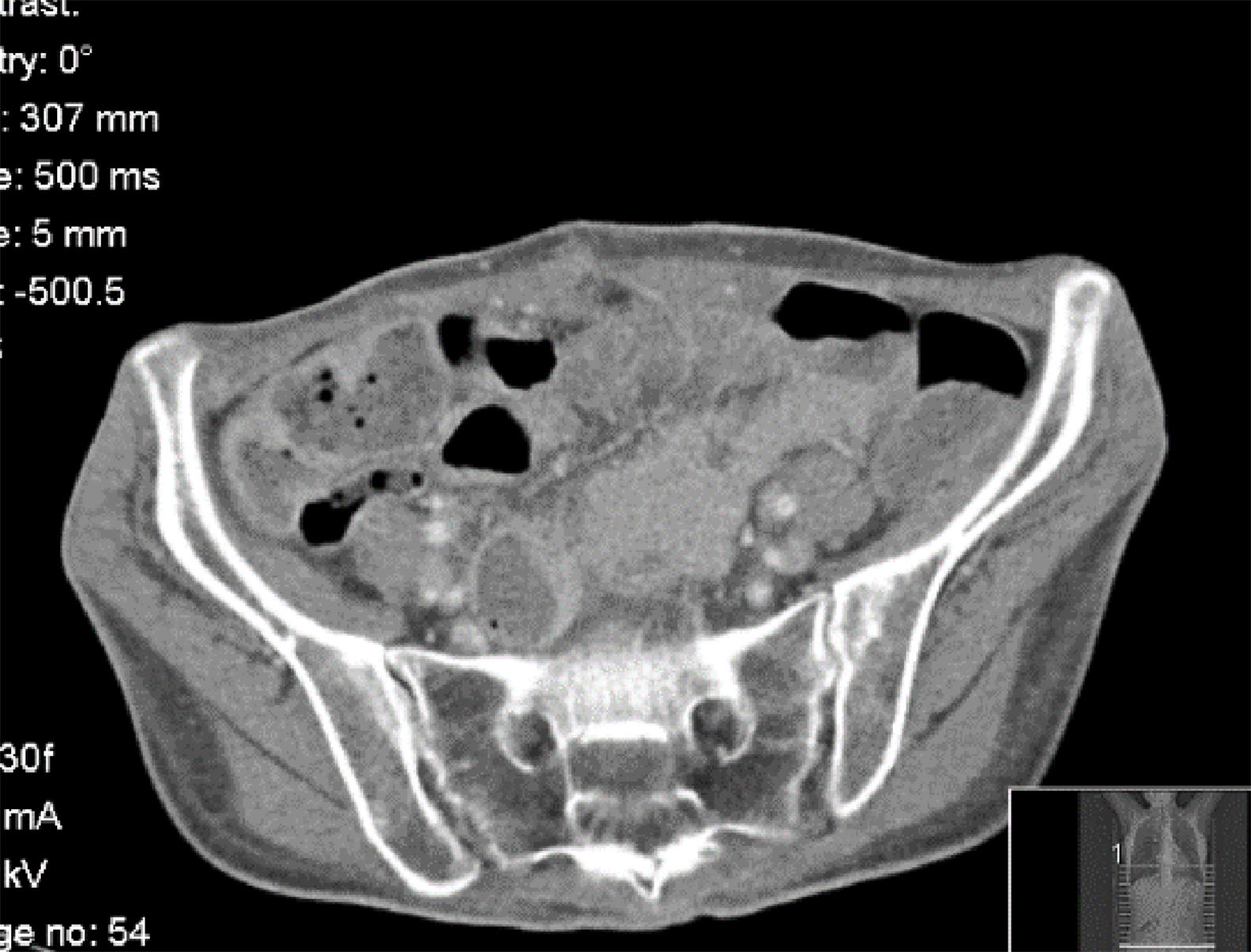
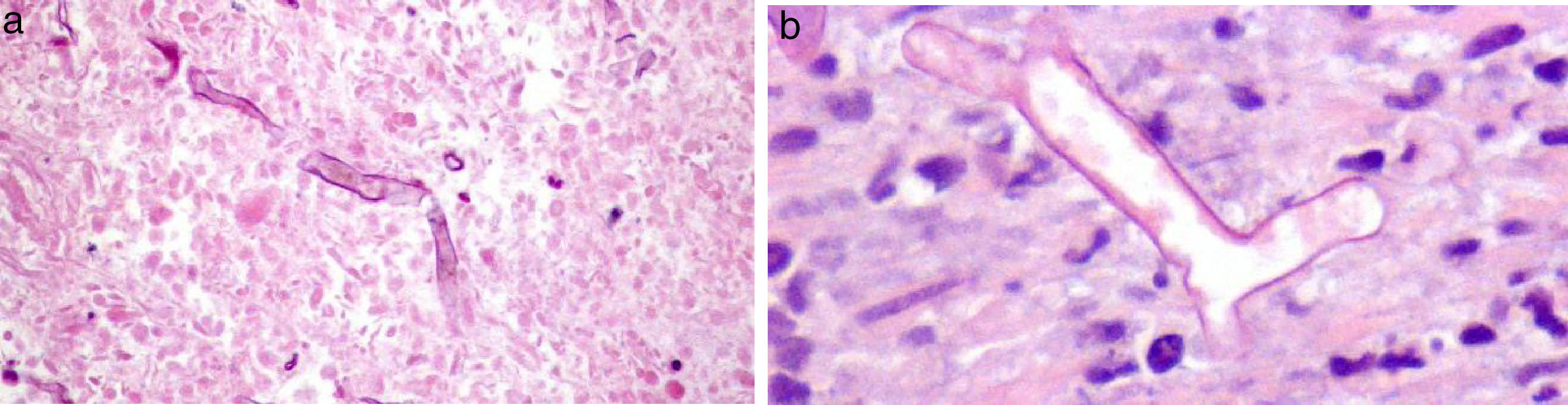
In the third month after transplantation, she complained of diffuse abdominal pain, with normal intestinal transit. Aerocolia in the terminal ileum and a slight thickening of the wall of the ascending colon were identified on abdominal ultrasound. Distal Intestinal Obstruction Syndrome (DIOS) was assumed, pro-kinetics drugs, oral rehydration and osmotic laxatives were prescribed and she was discharged. Forty-eight hours later, she was hospitalised due to non-resolving complaints (crampy diffuse abdominal pain). On day 2, during physical examination, an abdominal mass was identified in the right inferior abdominal quadrant. She was submitted to an abdominal computed tomography (CT) scan showing thickening of the wall of the ascendant colon and abnormal contrast accumulation, raising suspicion of an anterior intestinal wall perforation, despite no evidence of free air/fluid collection in the abdominal cavity (Fig. 1). A right hemicolectomy and reconstruction were performed without complications. The anatomopathological examination of the intestinal tissue showed necrotic areas and the presence of wall and vascular invasion by fungal hyphae, with distinction between Mucor or Aspergillus impossible on observation (Fig. 2a and b). The patient started empiric antifungal therapy with amphotericin, and switched to voriconazole after a positive test of polymerase chain reaction for Aspergillus.
Other infectious foci were excluded, namely lungs and heart. Abdominal fluid lavage, performed during surgery, blood cultures, coprocultures and Cytomegalovirus antigenemia were also negative. Six weeks later she did a control colonoscopy which showed no lesions. The patient suspended fungal therapy after 12 weeks and maintains regular follow up without intestinal complaints since treatment (5 years).
CF is generally thought to affect predominantly the lungs and is one of the main indications for lung transplant. Nevertheless, it is a systemic disease, with microbiologic diversity, and some symptoms or complications presented by these patients reflect these features.2
Gastrointestinal complications have become an increasing cause of morbidity in CF patients, in part due to better life expectancy. The mutations in transmembrane conductance regulator gene located on chromosome 7 are responsible for the abnormal chloride transport. The impaired chloride absorption is associated with tenacious mucus and secretions in the gut, pancreas and hepatobiliary system, alongside the lungs. Gastrointestinal manifestations of CF can be grouped into three categories: intestinal, pancreatic and hepatobiliary. The intestinal abnormalities include gastroesophageal reflux disease, meconium ileum, DIOS, intussusceptions, small bacterial overgrowth, constipation (defined as gradual faecal impaction of total colon) and rectal prolapse.3
DIOS, or meconium ileum equivalent, is characterized by an acute complete/partial obstruction of the ileum/cecum by intestinal contents, can occur at any age (although more common in older patients), in those with pancreatic insufficiency, with poorly controlled fat malabsorption and dehydration and also in transplanted patients.3–5 It occurs with acute onset of crampy abdominal pain, usually located in the right lower quadrant, with/without constipation. Sometimes an abdominal mass can be palpated, as in the case above. Differential diagnosis are appendicitis, volvulus or ovarian conditions. When DIOS is suspected, a CT scan can be performed usually showing proximal small-bowel dilatation and inspissated faecal material in the distal ileum; the absence of contrast material in terminal ileum is also suggestive. A water-soluble contrast enema can also be useful.
Regarding our case, diagnosis of DIOS was assumed as the clinical features and ultrasound findings were suggestive. But our patient was not just a CF patient but also a transplant recipient, who was under an intense immunosuppressive treatment.
It seems that immunosuppression can modify the normal intestinal flora, with bacterial and fungal overgrowth (namely Aspergillus), sometimes with transmural infarction and usually without evidence of systemic disease.6 The association of these two factors (DIOS and immunosuppression) probably explain the clinical findings, as both can be associated with changes in quality and quantity of the normal intestinal flora.
Initially, the patient started amphotericin with good tolerance. When Aspergillus was confirmed, it was switched to voriconazole.7 Besides the impossibility of drug monitoring of voriconazole,8 another question arose: the optimal duration of treatment. We decided to treat for 12 weeks after surgical resection and no lateral effects, intolerance or evidence of recurrence of the disease occurred.
The mortality rate of invasive aspergillosis in transplant recipients ranges from 65% to 92%, and 9.3 to 16.9% of all deaths in transplant recipients in the first year have been attributed to invasive aspergillosis.9 In the literature, we only found information about this diagnosis in necropsy samples, and there is also very little information about diagnosis, treatment or prophylaxis. These numbers are impressive, and they influenced our decision to share and discuss this case.
Conflicts of interestThe authors have no conflicts of interest to declare.