Moderate to severe pulmonary infectious exacerbations (PIE) are the main cause of admission in patients with cystic fibrosis (CF).1 Patients with non-CF bronchiectasis, as well as lung transplant recipients (LTR), are also often admitted for the same reason.1
Home intravenous antibiotic therapy (HIAT) has been used in the treatment of exacerbations, mainly in patients with CF, and additionally for the eradication of certain microorganisms such as Pseudomonas aeruginosa, with good tolerance and outcomes similar to inpatient intravenous antibiotic therapy (IAT).2 A multidisciplinary team (doctor, nurse, pharmaceutical) must be involved in order to select the patients and prescribe the best antibiotic, prepare the drugs and provide all care needed.2,3
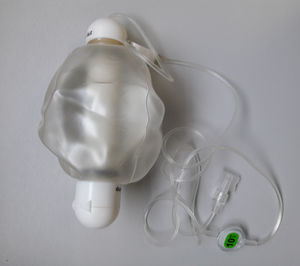
We aim to describe the experience of the first 20 months of HIAT in a Pulmonology Department of a tertiary hospital, using an elastomeric infusion pump Easypump® (Fig. 1).
All adult patients with CF, non-CF bronchiectasis or LTR who met the following inclusion criteria: (1) PIE with indication for admission or eradication of P. aeruginosa; (2) good adherence to previous prescribed medication (self-reported); (3) good family support; (4) home distance <2h from hospital; (5) good housing conditions (self-reported); (6) showed willingness to perform HIAT, were prospectively selected for it. Patients who presented respiratory failure at admission, sepsis or large volume haemoptysis were excluded. All patients were admitted in inpatient ward for at least 48h before starting HIAT, to provide instructions and training to the patients on how to use it safely at home, as well as explain possible complications inherent to its use, and document their clinical stability, to ensure a safe discharge home to complete treatment. A brochure with instructions and contact number were provided for the patients to use in case of doubt or complications. This kind of treatment was first approved by the hospital pharmacy and therapeutic committee and later by the hospital administration.
Eighteen courses of HIAT were run (two patients underwent the treatment twice). The median age was 32.5 years (minimum (min) 22 | maximum (max) 78) and sex distribution was equal.
Eight patients had CF, seven non-CF bronchiectasis, and one was a LTR with chronic rejection. One CF patient and one non-CF bronchiectasis patient were on the waiting list for transplant. Functionally, the patients had median FVC 68.5% predicted (min 46.7 | max 97.3), mean FEV1 39% predicted (min 21.8 | max 78.4) and median FEV1/FVC 48.3% (min 24.4 | max 69.9).
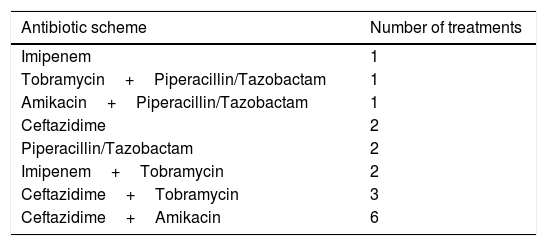
Twelve patients underwent HIAT due to PIE; 9 of them had chronic infection of P. aeruginosa, and the remaining for eradication of the same microorganism. The median time of admission was 6.5 days (min 2 | max 14), median time of HIAT was 9 days (min 6 | max 18) and median time of total treatment was 14 days (7 patients 21 days, 11 patients 14 days). The most commonly used antibiotic scheme was ceftazidime with amikacin, which was used in 6 patients (Table 1). Only one patient was unable to complete this antibiotic scheme after toxic levels of amikacin were detected. The most common complication was the blockage of peripheral venous lines. No adverse events or allergies related to HIAT occurred.
Only one CF patients was admitted in order to attempt eradication of P. aeruginosa. In these patients, statistically significant differences were found between median FVC % predicted (63.3% to 67.9%, p=0.034) and median FEV1% predicted (40.1% to 45.2%, p=0.049), before and after the HIAT. There was also improvement of FEV1/FVC (54.4% to 58.1%) but without statistical significance (p=0.123). Wilcoxon signed-rank test was used.
Despite the fact that this paper was not designed to evaluate cost-differences (due to methodology limitation), we were able to make an estimate of the costs. According to the prices stipulated by our hospital, taking into account the admission period and different IAT schemes, the total gross estimated price would have been 75761.2€ (mean 4209.0€/episode) if the treatment was carried out just in the hospital. The total gross estimated cost for our patients (admission, IAT, HIAT consumables and preparation) was 33102.9€ (mean 1839.1€/episode), saving in total 42658.3€. These costs did not include other drugs in addition to IAT that might have been used during admission (nebulization drugs, for example), the cost of blood analysis after discharge (at most 2 per patient) or other indirect costs such as loss of productivity, amongst others.
Several elastomeric infusion pumps are available for this kind of treatment. The Easypump® works independently of gravity or electricity. It can be stored at room temperature or refrigerated for several days, according to the drug to be administered.
Despite the fact that not all antibiotics can be administered through HIAT due to their stability characteristics, it has many advantages: it improves quality of life of patients, decreases absenteeism (work and school), admission days and the risk of nosocomial infections and provides cost savings, while maintaining equal treatment effectiveness.2,4–6
Although only a few patients have been enrolled so far, our preliminary experience with HIAT was very positive, with the CF patients achieving an improvement of their lung function, and only one patient not being able to complete treatment. However, not all patients are good candidates for this kind of treatment, so for HIAT to be successful it is necessary to carefully select patients and provide a well prepared multidisciplinary team for HIAT. An appropriate study to determinate the real cost differences is also needed.
Conflicts of interestThe authors have no conflicts of interest to declare.